Article II of the Chemical Weapons Convention (CWC) broadly defines a toxic chemical as any compound “that can cause death, temporary incapacitation or harm to humans or animals [1].” Figure 1 shows the five major classes of toxic chemical agents outlined by the CWC. With the exception of riot control agents used by law enforcement and the anti-proliferative properties of certain nitrogen mustards [2], many of these substances have no medical use and their weaponization is banned by the 193 nations committed to the Convention.
However, a potentially new class of chemical weapons is beginning to emerge—synthetic opioids. In 2017, opioids were responsible for more than 47,000 deaths in the U.S. and there is growing concern regarding their use in next-generation chemical weapons [3].
Apprehension is felt at the national and international level with the U.S. Department of Homeland Security (DHS) positioned to designate fentanyl a weapon of mass destruction and China pledging to halt the production of synthetic opioids altogether. Herein, we discuss the therapeutic properties of opioids, their pharmacology and toxicity, and the potential misuse of synthetic opioids in the theater of war.
Medical Use: A Double-Edged Sword
Opioids are highly addictive analgesics widely used in the management of moderate to severe pain. The Centers for Disease Control and Prevention (CDC) estimates that in 2017, 16% of U.S. counties saw enough opioids prescribed for every person to have a prescription [4]. These include a handful of natural, synthetic, and semi-synthetic compounds, such as morphine, oxycodone, codeine, tramadol, fentanyl, and hydrocodone. The pervasive use of opioids in the U.S. healthcare system distinguishes them from other chemical agents shown in Figure 1. Opioids offer many advantages over otheranalgesic drugs, namely superior pain relief, high central nervous system (CNS) permeability, a long duration of action, few cardiovascular risks, and a fast onset time.
Despite these therapeutic advantages, opioids produce fatal side effects when administered at the incorrect dose, echoing the onerous toxicology adage “the dose makes the poison.” This is particularly true for highly potent synthetic opioids like fentanyl and its derivatives (shown in Figure 2). A lethal dose of fentanyl is approximately 2 mg—the equivalent of a few grains of salt (see Figure 3) [5].
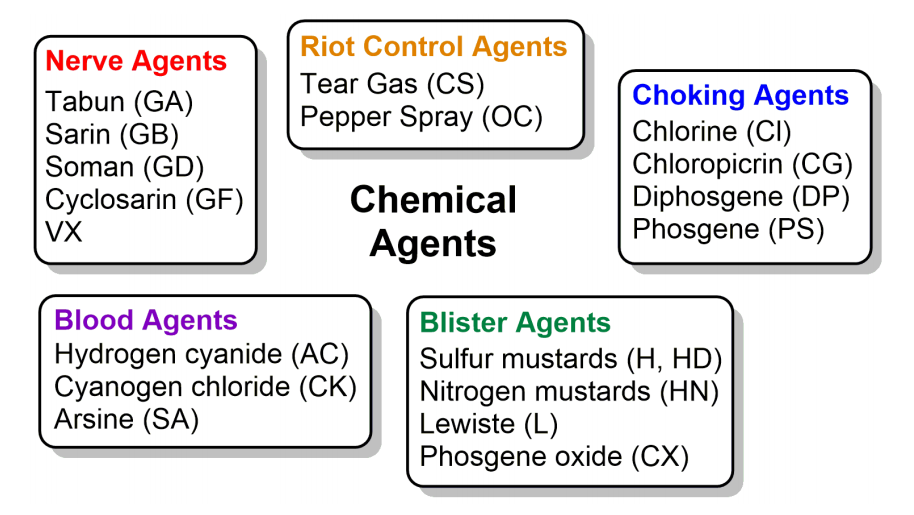
Figure 1. The five major classes of chemical agents identified by the Chemical Weapons Convention with examples listed in each group.
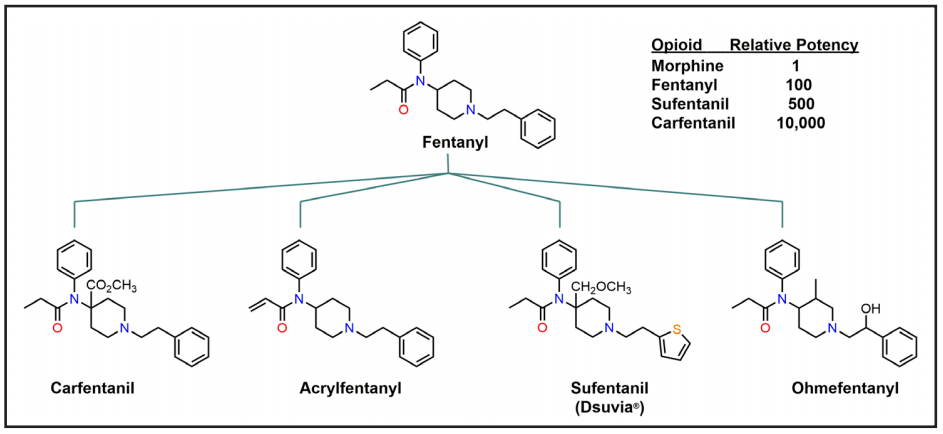
Figure 2. Many synthetic opioids are derivatives of fentanyl and are significantly more potent than morphine.

Figure 3. A lethal dose of fentanyl. Fentanyl is approximately 100-fold more potent than morphine and boasts an LD50 of 0.03 mg/kg in monkeys [5]. (Source: Drug Enforcement Administration)
Opioid Pharmacology and Mechanism of Toxicity
The on-target toxicity of opioids is inherently tied to the complexity of opioid signaling. Agonist opioids like fentanyl produce analgesia upon binding to their cognate receptors located throughout the central and peripheral nervous systems. Of the three classical opioid receptors (µ, κ, δ) it is thought that the activation of the µ-opioid receptor (MOR) in the brain is the primary mechanism of opioid-induced analgesia [6]. However, receptor activation is tightly regulated by β-arrestin2 to control the extent and duration of opioid signaling after the receptor binds to its
target [7]. Regulation by β-arrestin2 does not produce analgesia, but rather “arrests” opioid signaling and triggers a series of downstream signaling events that briefly remove the receptor from the cell surface. The temporary disappearance of MORs within the brain creates a negative feedback loop that results in tolerance, requiring more opioids to produce analgesia with the remaining receptors. However, if additional opioids are administered beyond the therapeutic dose, the remaining receptor population also becomes compromised, further dampening MOR
signaling throughout the CNS which leads to significant respiratory suppression and subsequent death [8]. Indeed, this is the primary mechanism behind opioid-related overdose fatalities and is the very property that transforms therapeutic opioids into potential chemical weapons.
Weaponization of Synthetic Opioids
The idea of opioid-based chemical weapons dates back to the discovery of fentanyl in 1960. Fentanyl and several fentanyl analogues were the focus of extensive U.S. military research from the 1970s through the 1990s, including inhalation studies on non-human primates to develop novel incapacitating agents “for situations where a quick knock-down agent is needed [9].” These efforts led to the discovery of sufentanil and carfentanil, two of the most potent synthetic analogues known, with respective potencies of 500x and 10,000x that of morphine (see Figure 2). Carfentanil causes severe respiratory depression and death in non-human primates at doses ranging from 2–14 µg/kg and its aerosolization may be more toxic than the nerve agent VX [10]. In 2016, the appearance of carfentanil in illegal markets prompted the U.S. Drug Enforcement Administration to issue an explicit warning stating that the drug is a serious danger to public safety, law enforcement, and healthcare personnel [11]. Carfentanil was responsible for approximately 11% of all opioid-overdose deaths in ten states from June 2016 to July 2017 and has a high potential for weaponization [12].
Most U.S. programs relating to the weaponization of synthetic opioids were terminated shortly before the U.S. joined the CWC in 1993; however, research in other countries continued [9]. In 2002, Russian authorities used an undisclosed mixture of aerosolized synthetic opioids believed to contain carfentanil (dubbed Kolokol-1) to counter forty insurgents during the Moscow theater crisis [13]. All forty insurgents were killed along with 204 unsuspecting hostages after exposure to the Kolokol-1 aerosol, revealing the potential use and limitations associated with synthetic opioids as non-lethal incapacitating agents.
The lethal consequences of Kolokol-1 did not deter Russian authorities from using synthetic opioids to counter other domestic terrorist groups. In 2005, Russian Special Forces descended on the town of Nalchik and subsequently deployed another unknown aerosol agent against militants holding hostages in a local shop. It is believed that the aerosol contained carfentanil and remifentanil, similar to the Kolokol-1 cocktail used in the Moscow incident [14]. Later that year, Russian researchers announced, but did not fully disclose, progress toward the development
of additional incapacitating agents at the 3rd European Symposium on Non-Lethal Weapons [15].
It’s unclear if these programs remain in operation today or if Russia has a continued interest in weaponizing synthetic opioids. However, growing precedent suggests that Russia is actively trying to obstruct CWC provisions relating to chemical weapon investigations in a likely attempt to shroud their motives, increasing political tensions between Moscow and the West [16].
To the best of our knowledge, there are no additional reports disclosing the use of weaponized opioids in acts of terror or on the battlefield at the time of this writing. Nonetheless, concerns are mounting. In January 2019, Massachusetts senator Ed Markey contacted Secretary of State Mike Pompeo and Acting Secretary of Defense Patrick Shanahan asking if their departments have “devised strategies to deal with the potential weaponization of fentanyl [17].” Fentanyl was responsible for nearly 28,000 overdose deaths in 2017 [18]. In response, DHS recently announced that it was considering classifying fentanyl as a weapon of mass destruction [19]. The announcement seeks to increase international awareness and the severity of repercussions to those harboring and distributing large quantities of fentanyl.
China has been the main supplier of fentanyl in the U.S., Canada, and Mexico, although it’s unclear if radical terrorist groups or nations have taken steps to exploit China’s supply for nefarious means [20]. Interestingly, China revealed a nation-wide ban on the production of synthetic opioids in April 2019, shortly before DHS announced its position on fentanyl [21]. The cross-talk between China and the Trump Administration represents a clear and coordinated effort to restrict the flow of synthetic opioids into illicit markets and to minimize potential security threats
that may arise from the weaponization of China’s fentanyl supply.
Antidotes
Naloxone hydrochloride (Narcan®) is the most pervasive antidote on the market and is widely distributed to emergency medical services (EMS) at the forefront of the opioid epidemic. Originally developed in the 1960s, naloxone shares many structural features with morphine (see Figure 4); however, subtle differences in structure give rise to significantly disparate properties. Unlike morphine, naloxone is a competitive MOR antagonist that effectively dislodges opioids from the receptor to reverse opioid-induced toxicity, specifically respiratory suppression.
The efficacy of naloxone has been extensively reviewed and depends on several key factors, including the route of administration, dose, and the identity of the opioid that is to be countered [22]. Naloxone has limited oral efficacy and is typically administered as an intravenous, intramuscular, or subcutaneous injection to circumvent its poor oral bioavailability and extensive first-pass metabolism. Complementary intranasal formulations are also available that have a rapid onset time and eliminate the use of needles [23]. In 2019, the U.S. Food and Drug Administration (FDA) approved the first generic formulation of naloxone hydrochloride nasal spray for use in a community setting by individuals without medical training [24]. Increased access to a user-friendly antidote will likely lessen the burden on EMS personnel and place more control in the hands of the public to mitigate the opioid epidemic. The technology may also find significant value in protecting civilians against a potential opioid-based chemical attack.
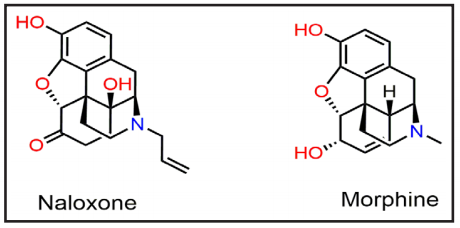
Figure 4. The chemical structures of naloxone and morphine share many structural similarities. However, naloxone is an antidote whereas morphine is an opioid.
Next-Generation Medical Countermeasures
Auto-injectors containing 0.4 mg of naloxone hydrochloride (Evzio®) and Narcan® nasal spray (2 mg/mL) sufficiently reverse respiratory suppression from most overdoses involving heroin and morphine; however, these doses are inadequate for potent synthetic opioids [25]. A recent report by Sommerville et al. found that 83% of suspected fentanyl overdoses in Massachusetts from 2014–2016 required more than two doses of Narcan® nasal spray before any resuscitative response was observed [26]. The same study also found that of the fatalities examined, 36% overdosed within minutes after fentanyl ingestion and 90% of these patients were pulseless upon EMS arrival. These findings demonstrate how quickly opioid-induced respiratory suppression progresses to death and is a stirring reminder of the Moscow theater hostage crisis where synthetic opioids proved to be more than non-lethal incapacitating agents. The potency of fentanyl and its derivatives calls into question the proper dose of naloxone that should be administered to rescue patients in the era of synthetic opioids.
Single-use, high potency antidotes that work on a majority of the population are critical in protecting the public from synthetic opioids and mitigating the effects of a domestic chemical attack. In 2016, the FDA approved a new formulation of Evzio® that contains 2mg of naloxone hydrochloride—a dose five times higher than the original device. However, a high potency antidote doesn’t necessarily translate to single use. This is especially true for lipophilic opioids that leach out from adipose tissues and agonize the MOR for long periods of time.
To address this concern, researchers at the American Chemical Society National Meeting & Exposition (Spring 2019) announced the development of a next-generation single-dose opioid antidote using naloxone-loaded polylactic acid nanoparticles that slowly release therapeutic concentrations of naloxone over 24 hours [27]. No results are available at this time and it remains to be seen if this technology will pave the way to single-use antidotes against synthetic opioids. In a similar vein, the U.S. Department of Health and Human Services is sponsoring the development of a long-lasting intranasal formulation of naloxone to circumvent the need for multiple doses [28].
Additionally, DHS Science & Technology Directorate (DHS S&T) is working to protect first responders at risk of exposure to harmful levels of fentanils at emergency scenes.
DHS S&T partnered with Clear Scientific to create ClearAlert—a wearable sensing device that aims to quickly alert first responders to their level of exposure to fentanils, thus allowing them to rapidly exit the toxic environment and receive medical countermeasures necessary to prevent serious injury [29]. According to DHS S&T, “Experimental results indicated successful and rapid detection of submicrogram quantities of aerosolized carfentanil [29].” Future development of the device will focus on sensor performance and operational life optimization, and field testing
and market research will be performed to ensure the final product meets the requirements of the first responder community.
Conclusion
Synthetic opioids represent a unique class of potential chemical weapons with significant medical use. Historical evidence suggests that weaponized opioids are not the preferred agent of choice for radical states or nations; however, Russia’s newfound defiance over CWC provisions and previous use of Kolokol-1 makes the future of chemical peace unclear. The Trump administration and members of Congress recognize the weaponization potential of synthetic opioids and have taken steps to potentially classify fentanyl as a weapon of mass destruction [19]. In addition, China’s pledge to ban the production of synthetic opioids will significantly limit fentanyl distribution in underground markets and hinder illicit stockpiling by bad actors [20-21].
As DHS aims to control the flow of fentanyl overseas, the FDA continues to expedite approvals for novel naloxone formulations to combat the opioid epidemic. New technologies such as user-friendly nasal aspirators and single-use naloxone antidotes circumvent the need for extensive medical training and provide the public with direct access to medical countermeasures that may prove invaluable during a potential chemical attack with synthetic opioids.
References
1. Defense Threat Reduction Agency. (2002). Chemical Weapons Convention. Retrieved from https://www.opcw.org/chemical-weapons-convention/articles/article-ii-definitions-and-criteria
2. Singh, R. K., Kumar, S., Prasad, D. N., & Bhardwaj, T. R. (2018). Therapeutic journery of nitrogen mustard as alkylating anticancer agents: Historic to future perspectives. European Journal of Medicinal Chemistry, 151, 401-433. doi:10.1016/j.ejmech.2018.04.001
3. Scholl, L., Seth, P., Kariisa, M., Wilson, N., & Baldwin, G. (2019). Drug and opioid-involved overdose deaths—United States, 2013–2017. Morbidity and Mortality Weekly Report, 67(5152), 1419.
4. Centers for Disease Control and Prevention. (2018). U.S. opioid prescribing rate maps. Retrieved from https://www.cdc.gov/drugoverdose/maps/rxrate-maps.html
5. Burns, S. M., Cunningham, C. W., & Mercer, S. L. (2018). DARK classics in chemical neuroscience: Fentanyl. ACS Chemical Neuroscience, 9(10), 2428-2437. doi:10.1021/acschemneuro.8b00174
6. Pathan, H., & Williams, J. (2012). Basic opioid pharmacology: An update. British journal of pain, 6(1), 11-16. doi:10.1177/2049463712438493
7. Raehal, K. M., & Bohn, L. M. (2014). β-arrestins: Regulatory role and therapeutic potential in opioid and cannabinoid receptor-mediated analgesia. Handbook of Experimental Pharmacology, 219, 427-443. doi:10.1007/978-3-642-41199-1_22
8. Boom, M., Niesters, M., Sarton, E., Aarts, L., Smith, T. W., & Dahan, A. (2012). Non-analgesic effects of opioids: Opioid-induced respiratory depression. Current Pharmaceutical Design, 18(37), 5994-6004.
9. Pearson, A. (2006). Incapacitating biochemical weapons. The Nonproliferation Review, 13(2), 151-188. doi:10.1080/10736700601012029
10. Stanley, T. (2003). Human immobilization: Is the experience in Moscow just the beginning? European Journal of Anaesthesiology, 20(6), 427-428.
11. U.S. Drug Enforcement Administration. (2016). DEA issues carfentanil warning to police and public. Retrieved from https://www.dea.gov/press-releases/2016/09/22/dea-issues-carfentanil-warning-police-and-public
12. O’Donnell, J., Gladden, R. M., Mattson, C. L., & Kariisa, M. (2018). Notes from the field: Overdose deaths with carfentanil and other fentanyl analogs detected—10 states, July 2016–June 2017. Morbidity and Mortality Weekly Report, 67(27), 767.
13. Riches, J. R., Read, R. W., Black, R. M., Cooper, N. J., & Timperley, C. M. (2012). Analysis of clothing and urine from Moscow theatre siege casualties reveals carfentanil and remifentanil use. Journal of Analytical Toxicology, 36(9), 647-656. doi:10.1093/jat/bks078
14. Davison, N., & Lewer, N. (2006). Bradford non-lethal weapons research project (BNLWRP). Research Report No. 8. Retrieved from University of Bradford, Department of Peace Studies, Centre for Conflict Resolution: http://hdl.handle.net/10454/4000
15. 3rd European Symposium on Non-Lethal Weapons. (2005). Retrieved from http://www.non-lethal-weapons.com/sy03index.html
16. Sanders-Zakre, A. (2019, February). Russia blocks consensus at CWC conference. Arms Control Today. Retrieved from https://www.armscontrol.org/act/2019-01/news/russia-blocks-consensus-cwc-conference
17. Edney, A. (2019, January 28). Senator seeks strategy to prevent fentanyl terror attacks. Bloomberg. Retrieved from https://www.bloomberg.com/news/articles/2019-01-28/strategy-to-prevent-fentanyl-terror-attacks-sought-by-senator
18. Centers for Disease Control and Prevention. (2018). Overdose deaths involving synthetic opioids. Retrieved from https://www.cdc.gov/drugoverdose/data/fentanyl.html
19. Turner, A. (2019, April 16). Homeland security is repordetly considering labeling fentanyl as a weapon of mass destruction. CNBC. Retrieved from https://www.cnbc.com/2019/04/16/dhs-considering-labeling-fentanyl-as-weapon-of-mass-destruction-report.html
20. U.S. Drug Enforcement Administration. (2016). Counterfeit prescription pills containing fentanyls: A global threat. Retrieved from https://content.govdelivery.com/attachments/USDOJDEA/2016/07/22/file_attachments/590360/fentanyl%2Bpills%2Breport.pdf
21. Myers, S. G., A. (2019, April 1). China bans all types of fentanyl, cutting supply of deadly drug to U.S. and fulfilling pledge to Trump. The New York Times. Retrieved from https://www.nytimes.com/2019/04/01/world/asia/china-bans-fentanyl-trump.html
22. Rzasa Lynn, R., & Galinkin, J. L. (2018). Naloxone dosage for opioid reversal: Current evidence and clinical implications. Therapeutic advances in drug safety, 9(1), 63-88. doi:10.1177/2042098617744161
23. Robinson, A., & Wermeling, D. P. (2014). Intranasal naloxone administration for treatment of opioid overdose. American Journal of Health-System Pharmacy, 71(24), 2129-2135. doi:10.2146/ajhp130798
24. U.S. Food and Drug Administration. (2019). FDA approves first generic naloxone nasal spray to treat opioid overdose. Retrieved from https://www.fda.gov/news-events/press-announcements/fda-approves-first-generic-naloxone-nasal-spray-treatopioid-overdose
25. Moss, R. B., & Carlo, D. J. (2019). Higher doses of naloxone are needed in the synthetic opioid era. Substance Abuse Treatment, Prevention, and Policy, 14(1), 6. doi:10.1186/s13011-019-0195-4
26. Somerville, N. J., O’Donnell, J., Gladden, R. M., Zibbell, J. E., Green, T. C., Younkin, M., … Walley, A. Y. (2017). Characteristics of fentanyl overdose – Massachusetts, 2014-2016. MMWR: Morbidity and Mortality Weekly Report, 66(14), 382-386. doi:10.15585/mmwr.mm6614a2
27. Cottingham, K. (2019). Next-generation single-dose antidotes for opioid overdoses. Retrieved from https://www.acs.org/content/acs/en/pressroom/newsreleases/2019/april/next-generation-single-dose-antidotes-for-opioid-overdoses.html
28. U.S. Department of Health and Human Services. (2018). HHS sponsors development of intranasal form of long-acting opioid overdose drug. Retrieved from https://www.hhs.gov/about/news/2018/09/20/hhs-sponsors-development-intranasal-form-long-acting-opioid-overdose-drug.html
29. Department of Homeland Security. (2019) ClearAlert: Wearable sensing badge for solid aerosol and contact exposure to fentanils. Retrieved from https://www.dhs.gov/sites/default/files/publications/dhs_clearalert_onepager_final-508.pdf


