There is an overwhelming need to conduct diagnostic tests on soldiers deployed in remote locations, where a clinical laboratory is not available. To assess, for example, the extent of head trauma and brain injury; to identify whether an infection is due to dengue or malaria; to determine if illness is caused by a pathogen such as anthrax or other chemical or biological agents; to check for drug use; or to simply assess the soldiers’ wellness. Early and timely detection of agents used to cause significant harm is critical to assisting the warfighter.
The ability to conduct meaningful tests at the doctor’s office, at the pharmacy or home, at the scene of an emergency or in the ambulance on the way to the hospital has many benefits. One outcome could be lower health care costs because a doctor can make a diagnosis while the patient is still in his office. Testing at the pharmacy saves on doctor’s visits. If a patient can be tested by emergency medical technicians, it will save time and possibly the person’s life if the doctors in the emergency room know the test results. Doctors can be better prepared and may begin treatment, such as surgery, right away rather than wait to conduct relevant tests after the patient arrives at the hospital.
This need exists also for people who live in isolated communities around the world or even in space. With present day telecommunications, a test could be conducted locally and diagnosis made by a health professional located far away.
To meet this need, considerable effort has been expanded over the past 30 years on a class of devices called lateral flow assays. The term lateral flow serves to point out how the various fluids in these devices flow laterally, as opposed to the conventional diagnostic systems where the various reagents are added and removed vertically. There are two types of lateral flow assay devices: polymer-based and paper-based. Polymer-based devices have received excellent reviews. [1,2] Opko Health, Inc. [4] is commercializing a system based on this technology. A very comprehensive review of paper-based devices is offered in Lab on a Chip Journal. [3] Chembio, Inc. [4] is selling paper-based devices. In addition, there are a great number of companies selling home pregnancy test kits, which are the most successful of all lateral flow assays. [5]
Polymer-based lateral flow assays, despite their many apparent advantages, have not become commercially successful. There are a number of technical reasons for this. [6,7] The most important reason may be the power requirement and, the user must manipulate a number of buttons and other tasks for it to perform the test. In addition, they are not generally affordable by someone who wishes to conduct a test only once.
Paper-based devices, on the other hand, require no power because fluid flows naturally by capillarity. The user for a home pregnancy test kit, for example, simply loads the sample and reads the results about 20 minutes later. There are no buttons to push, no switches to flip on or off and, once finished, the test can be easily disposed (including by burning) to minimize contamination. The cost is low and affordable for a one time test. The major advantages of these paper-based devices, or test strips, as they are commonly known, are their low cost, ease of use, passive and autonomous operation and portability. The major shortcoming is that they can accept only one reagent, or sample fluid. Consequently, they are not able to conduct complex assays requiring multiple reagents and this limits their application space and sensitivity.
A new paper-based device maintains all the advantages of the current test strips but adds the ability to do more complex tests.
These new types of lateral flowdevices are based on the invention of a fluid-actuated microfluidic diode, or valve that is built in paper. [8,9,10] Figure 1 shows a schematic drawing of the diode. The left side shows a simplified cross-sectional view consisting of two layers of paper separated by a layer of double sided tape. A hole is punched in the tape ahead of time and two discs, both made of paper, are placed in the hole. The hydrophobic disc soaked in a solution containing Allyltrichlorosilane renders its fibers hydrophobic but the disk remains porous. The hydrophilic disc is infused with Tween 20 surfactant. A symbol of the paper-based microfluidic valve is shown in the right side of the figure.
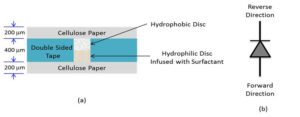
Figure 1: (a) Schematic cross-sectional view of a fluid actuated fluidic diode or valve. (b) Valve symbol. (Image courtesy of Constantine Anagnostopoulos and Mohammad Faghri/Released)
In operation, if the fluid approaches the diode from the forward direction, it dissolves the surfactant, which goes into the hydrophobic area and renders the fibers hydrophilic again. If the fluid approaches from the reverse direction, it faces a hydrophobic region and cannot penetrate it. However, unlike electrical diodes, once the diode is bridged, it allows fluid to flow in both directions.
A drawing of the top view of one paper-based lateral flow device utilizing fluidic diodes is shown in Figure 2. The white areas correspond to virgin paper. Wax has been deposited in the black regions and has been melted throughout the bulk of the paper forming a barrier to fluids. The green patch corresponds to a glass fiber membrane. The yellow test spot is defined in a white nitrocellulose membrane. The white membrane is barely visible in this figure.
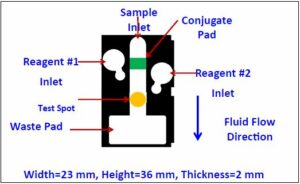
Figure 2: Top view of the new paper-based lateral flow device with fluid management. (Image courtesy of Constantine Anagnostopoulos and Mohammad Faghri/Released)
There are four distinct regions of interest: the central channel that begins at the sample inlet and ends at the waste pad, the two circular regions marked Reagent 1 Inlet and Reagent 2 Inlet; and the waste pad. Conventional test strips, like the home pregnancy test, consist only of the central channel. The device in Figure 2 is capable of conducting several different kinds of assays, including one of the most common diagnostic tests called Enzyme Linked Immunosorbent Assay. A schematic representation of an ELISA protocol for the detection of Rabbit IgG on our paper-based device is shown in Figure 3. An equivalent fluidic circuit of the chip of Figure 2 is shown in Figure 4. IgG stands for immunoglobulin G and is an antibody. In this case IgG was generated by a rabbit’s immune system. It is a benchmark biomarker, which is one that is used by many researchers so they can compare their results to others.
To conduct the Rabbit IgG test, first Reagents 1 and 2 are preloaded on their respective inlets. This can be done at the factory at the time the devices are made so the user does not have to load them at the time of the test. Reagent 1 is a wash solution, typically phosphate-buffered saline, which is a buffer solution commonly used in biological research. It is a water-based salt solution containing sodium phosphate, sodium chloride and, in some formulations, potassium chloride and potassium phosphate. Reagent 2 is the substrate BCIP/NBT. Also at the time the devices are made, detection antibodies are dried on the conjugate pad. The antibodies are monoclonal mouse anti-Rabbit IgG, which are specific to Rabbit IgG and are tagged with the enzyme Alkaline Phosphatase. And in the nitrocellulose membrane, the test spot is defined by placing a drop of fluid also containing monoclonal mouse anti-Rabbit IgG antibodies and allowed to dry. Because of the nature of the nitrocellulose membrane, these antibodies are immobilized in the spot where they were deposited.
At the time of the test, the user applies the sample at the sample inlet. Some of this fluid travels down the central channel where it picks up detection antibodies from the conjugate pad. The conjugates antigen+detection antibody+enzyme are formed and they continue flowing toward the test spot. As they flow over the test spot, the antigens are captured by the immobilized capture antibodies forming complexes, or sandwiches, of capture antibody+antigen+detection antibody+enzyme. This is demonstrated schematically in Step 3 on Figure 3.
A portion of the sample fluid flows in a channel below the surface, not visible in Figure 2, but shown as Delay Channel 1 of Figure 4. It reaches below Reagent 1, turns on the valves and activates its flow. In the assay described in Figure 3 this reagent is a simple wash solution. It flows into the central channel and its function is to remove any of the conjugates not properly anchored in the test spot, which is shown schematically as Step 4 in Figure 3. A portion of the sample fluid and wash solutions flow via another channel below the surface, designated as Delay Channel 2 in Figure 4, reaches Reagent 2 and activates its flow. Reagent 2 is the substrate and its function is to interact with the ALP enzymes that are on top of the complexes in the test spot and produce colored molecules that precipitate near the test spot and are visible. These precipitates constitute the signal. This is Step 5 in Figure 3.
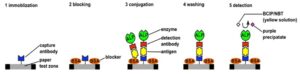
Figure 3: Schematic of the ELISA protocol for the detection of Rabbit IgG on our lab-on-paper device. (Image courtesy of Constantine Anagnostopoulos and Mohammad Faghri/Released)
By conducting this assay with samples containing different concentrations of Rabbit IgG, we have obtained a dose response or standard curve for our system. The curve is shown in the left side of Figure 5. The limit of detection was about 4.7ng/ml, which is similar to what is obtained in conventional ELISA tests using microtiter plates. The right side shows two chips at the end of the assay. In the top one, the test spot was obtained for an IgG concentration of 0.1μg/ml and the bottom one, the IgG concentration in the sample was 1μg/ml. The darkness of the spots correspond to the signal and it is seen that in the bottom chip the test spot is much darker than the spot on the top chip, as expected.
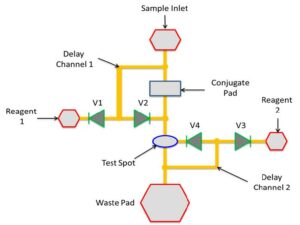
Figure 4: Equivalent fluidic circuit diagram for the device in Figure 2. (Image courtesy of Constantine Anagnostopoulos and Mohammad Faghri/Released)
The work reported above helped prove that these new devices can conduct ELISA autonomously. The ability to conduct assays autonomously is a big advantage when the tests are conducted outside a laboratory environment and by people who are not highly trained in laboratory procedures. In addition, ELISA is capable of higher sensitivity compared to test strips that use gold or other colored nanoparticles. This is because ELISA amplifies the signal since enzymes keep producing a color product as long as substrate molecules come in contact with them. The higher sensitivity means lower concentrations of an antigen can be detected. Finally, ELISA is a ubiquitous technique and has been used to detect many different kinds of molecules, including hydrocarbons or other contaminants in water, chemical warfare agents and more. It is expected that the standard ELISA protocols for these molecules can be transcribed for our paper-based devices, as it was shown schematically in Figure 3 for Rabbit IgG. Finally, ELISA provides not only yes/no results but quantitative data as well. This requires the color intensity of the test spots be measured and compared to the intensity in the appropriate standard curve, such as the one shown in Figure 5, from where the unknown concentration of an antigen or analyte in a sample can be determined. Presently, apps in smart phones or small units are capable of conducting these measurements with the added bonus that the data can be transmitted to health care or other professionals for evaluation or diagnosis.
To demonstrate that a conventional ELISA protocol can be transcribed for our paper-based devices, we collaborated with a local biotech start-up, ProThera Biologics, to detect a protein called Inter Alpha Inhibitor Protein (IαIP). [11] IαIPs are natural serine protease inhibitors found in human plasma at a relatively high concentration ranging between 400 – 800 αg/mL. IαIPs are part of the body’s protective mechanism that modulates host response to pathological insults. Circulating IαIP levels are significantly decreased in adult and neonatal sepsis. The total IαIP levels correlate inversely with the mortality rate in adult patients with severe sepsis. It is known that IαIPs, as part of the innate immune response, protect against acute, systemic inflammation following severe bacterial or viral infections, such as those resulting from septic shock or dengue shock syndrome. As a consequence, these proteins are rapidly consumed and excreted in the urine, leading to a rapid decrease in plasma levels. Moreover, in several adult and newborn animal models of sepsis as well as anthrax intoxication and infection, IαIP replacement therapy has been demonstrated to reverse the decrease in system levels, thereby significantly reducing sepsis related mortality. Since IαIP replacement therapy has been demonstrated to be beneficial in sepsis and anthrax infection, IαIP has great potential both as a predictive marker and therapeutic agent. [12] We have been able to produce a quantitative rapid test on our paper based devices that measures the concentration of IαIPs in buffer by either direct ELISA or competitive ELISA methods. [13, 14]
Further, the researchers at ProThera Biologics, under a grant from the Department of Defense, are currently investigating whether the concentration of IαIPs in the blood is reduced for people who have suffered a severe brain injury or stroke. If this proves to be the case, a rapid test for use in the field by the military would be extremely useful because it will alert the medical staff of the possible onset of sepsis. Another group of researchers [15] have identified biomarkers in the blood that can determine the level of brain damage a soldier or athlete may have suffered from a blow to the head. Up until now, concussion diagnosis has been limited to cognitive measures that can be subjective. These researchers are interested in developing a rapid test to measure the concentrations of the identified proteins within two hours or less of the injury so they may be able to treat the patient more effectively and thus minimize brain damage. The rapid test must provide quantitative results and our new paper-based devices can do that.
Knowing the concentration of inhibitors such as IαIPs, however, may not be sufficient. This is because in case of illness or injury the quality of IαIPs may be impaired. Thus, while an ELISA or another sandwich assay may detect a certain concentration of IαIPs in the sample, the number of these proteins that are actually active may be considerably less. Experiments are being conducted on dual devices, where one device will measure the concentration of these proteins in the sample while simultaneously, and from the same sample, the second device will measure the activity levels of these inhibitors.
A much broader field, where knowing at the same time the concentrations as well as the activity levels of enzymes and inhibitors is important, is in the areas of hemostasis and thrombosis. In these cases, enzymes help to form clots in the event of an injury to stop the bleeding while simultaneously inhibitors deactivate the enzymes to prevent them from forming large blood clots that could break off and result in thrombosis. These types of assays, however, cannot be done autonomously by the conventional paper-based lateral flow test strips because they can handle only one reagent while these tests require a minimum of two reagents, which our paper-based devices can manage easily.
Finally, one area of interest to the military is the accurate diagnosis of dengue virus infection. Dengue represents an ongoing global health problem, with an estimated 390 million infections per year and complicates deployment of U.S. military personnel to hot spots including Southeast Asia and the Middle East. [16] Substantial efforts have been made in the development of diagnostic tests for detection of dengue during the acute illness phase. Several commercial rapid diagnostic lateral flow assays are available for diagnostic testing outside the United States, mainly based on detection of the dengue virus NS1 protein in blood. Sensitivity of these commercial assays, however, is reported to be in the range of 60-80 percent, and is significantly lower in secondary dengue virus infections, which are associated with a higher risk of more severe disease. Further, current rapid diagnostic tests do not provide quantitative information to identify patients at higher risk for severe disease. The ability of these paper-based devices to conduct ELISA could meet the need of the military, and communities at large, for low cost, but higher sensitivity, rapid and quantitative diagnostic tests for dengue.
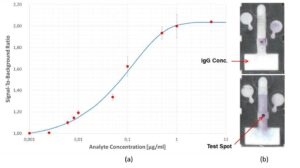
Figure 5: (a) Dose response curve for Rabbit IgG; (b) Photomicrographs of two chips after running ELISA. (Image courtesy of Constantine Anagnostopoulos and Mohammad Faghri/Released)
Acknowledgements:
This work was supported in part by NSF PIRE grant NSF-OISE-0530203 and a 2012 STAC grant from the State of Rhode Island. Also, the many contributions of Yow-Pin Lim, M.D./Ph.D. from ProThera Biologics are also gratefully acknowledged.
References:
[1] Chin, C., Laksanasopin, T., Cheung, Y.K., Steinmiller, D., Linder, V., Parsa1 H. … & Sia, S.K., (2012, July 31). Microfluidics-Based Diagnostics of Infectious Diseases in the Developing World. Nature Medicine. doi:10.1038/nm.2408
[2] Chin, C., Cheung, Y.K., Laksanasopin, T., Modena, M., Chin, S.Y., Sridhara, A.A. … & Sia, S.K. (2013). Mobile Device for Disease Diagnosis and Data Tracking in Resource-Limited Settings. Clinical Chemistry, 59:4, 629–640.
[3] Yetisen, A.K., Akram, M.S. & Lowe, C.R. (2013). Paper-Based Microfluidic Point-of-Care Diagnostic Devices. Lab Chip,13, 2210–2251.
[4] Claros Diagnostics, (now Opko Diagnostics); Chembio.
[5] Waddell, Rebecca S. (2006). Home Pregnancy Test hCG Levels and FAQ. Fertility Plus.
[6] Franzblau, Michael. (2014, December). Portable Microfluidic Fluid Handling System for the Detection of Protein Biomarkers in Whole Blood. (Master’s Thesis). University
About the Authors:
Constantine Anagnostopoulos, Ph.D., is an adjunct full professor at the University of Rhode Island working on lab-on-a-chip and lab-on-paper platform technologies. Before joining the microfluidic laboratory, he worked at the research and development laboratories of the Eastman Kodak Company, leading research teams focusing on inventing, maturing and finally moving ideas to products. He is on more than 70 U.S. patents and published more than 50 papers in peer-reviewed journals. He founded Labonachip, LLC to commercialize the lab-on-a-chip and lab-on-paper technologies.
Mohammad Faghri, Ph.D., is a professor of mechanical engineering at the University of Rhode Island. He is known for his work in microfluidics, lab-on-paper and lab-on-a-chip technologies. He has published more than 200 articles in peer reviewed journals and authored seven books. Dr. Faghri has received numerous awards for his scientific contributions as well as several grants. His research interest has been on bio-micro-electrical-mechanical systems with applications for development of a lab-on-a-chip and lab-on-paper devices for point-of-care diagnostics.


