Historically, burns have accounted for 5 to 20 percent of combat injuries, with 4 percent of burn victims succumbing to their injuries. According to the United States Army Institute for Surgical Research Burn Center, participation in Operation Iraqi Freedom (OIF) and Operation Enduring Freedom (OEF) have contributed to higher burn-injury rates than previously seen in other conflicts [1,2]. This increase is likely due to the use of improvised explosive devices (IEDs) by enemy combatants [1], which has not been experienced in past operations.
The burn potential and patterns inflicted by IEDs are unique and can be quite severe [3], making a significant contribution to the number of burns sustained by military personnel as the result of hostile action in areas of combat operations [4,5]. Burn injuries that occur in combat often result in a decrement in fighting strength and physical impairment, which may require rehabilitation or lead to lifelong disability and even death. In response to the increase in combat-related burn injuries, U.S. Army Natick Soldier Research, and Development and Engineering Center (NSRDEC), as part of its ongoing mission to protect and sustain the Soldier, continues to evaluate methods of developing new protective garments that can reduce the likelihood and severity of burns due to thermal threats on the battlefield.
Burn Injury Model
An essential aspect for evaluating protection against battlefield hazards lies in the means to predict the extent and severity of skin burns that can arise under specific thermal exposure conditions faced by the warfighter. As such, testing methods (e.g., ISO 17492 [6] and ASTM F1930 [7]) conducted at NSRDEC’s Thermal Test Facility routinely assess thermal protection afforded by fabrics and garments by incorporating a burn injury model developed by Stoll and Chianta [8-10].
Limitations
Although protective garments are designed for all areas of the body, burn prediction testing currently used in military lab settings is based entirely on limited human test data for superficial second-degree burns on the volar surface of the forearm (indicated by formation of a blister within 24 hours), which does not take into account the considerable differences in tissue properties and thicknesses (affecting burn damage response) across the body. It is routine in garment testing to predict areas of significantly greater burn injury severity (deeper second- and third-degree), however, this prediction is based on specific yet limited data. Additionally, anesthetized animals and human cadavers have historically been used to research burn severity. An improved whole body burn injury model is needed that incorporates appropriate body-region-specific behavior for the development, performance, and assessment of protective clothing.
Human skin is a complex tissue, consisting of multiple layers, vascular networks, and cell types that together create a barrier against environmental factors. Current models used to recapitulate skin include 2-D cell cultures and animals. 2-D cell cultures are the most widely
used skin models as they provide a simple, inexpensive system for modeling single layers of cells found in skin. However, 2-D cell cultures do not mimic key 3-D cell-extracellular matrix interactions, chemotactic cell migration, blood flow, and other physiologic factors present in skin. As a result, these models do not mimic the skin’s response to external stimuli, such as battlefield injuries, including burns, exposure to chemicals, and varying environmental stimuli (e.g., extreme temperature, moisture, and particulates), and contact with varying types of materials (e.g., protective garments). While animal models of skin provide better physiologic fidelity, they are expensive and do not fully replicate the human phenotype and its response. The development of an inexpensive, physiologically representative skin platform capable of characterizing skin response to battlefield injuries or severe environmental conditions in the presence or absence of protective materials could positively influence wound treatment
Current Research
NSRDEC and University of Texas at Austin researcher M. Nichole Rylander are collaborating to develop a representative human skin simulant having ‘true’ human skin cell layers and vasculature that will allow for a more relevant testing approach to burn injury when testing to radiant and convective heat fluxes. These vascularized tissue platforms incorporate simulated blood perfusion and biologic materials so that biochemical markers and cellular responses involved in burn injury may be directly observed. The objective of this research is to develop skin burn injury models capable of predicting a range of skin injury from threshold second-degree burns to severe (deep second- and third-degree) burns and to validate the models against available human and animal model experimental data.
Approach
Researchers at the University of Texas at Austin’s Rylander Lab have created a 3-D physiologically representative collection of skin platforms that range from single skin layers to complex, vascularized, full-thickness skin consisting of matrices prevalent in each skin layer: collagen (dermal layer) and keratin (epidermal layer). Researchers have created avascular single- and multi- layer skin with fibroblasts, keratinocytes, and endothelial cells (all cells present in the human skin) so injury may be studied independently for each layer (see Figure 1A).
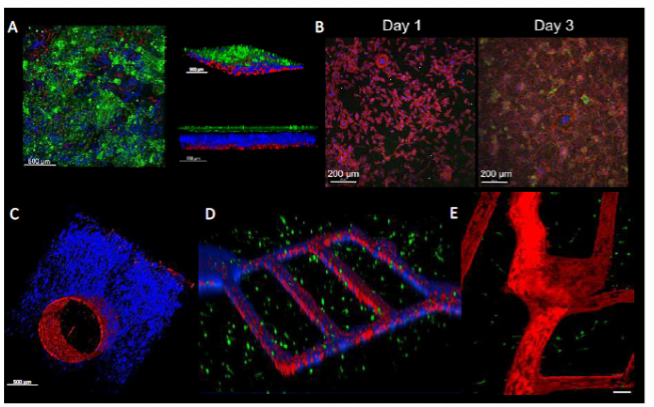
Figure 1: Skin platforms created by the Rylander Lab. (A) Full-thickness avascular skin platform flourescence images containing represetative cell types keratinocytes, fibroblasts, and endothelial cells
with angled view (top), side view, and top view below (B) Differentiated keratinocytes
expressing caspase 14 (green) after three days (C) Vascularized dermis with fibroblasts shown in blue and red endothelias cells lining a representative blood vessel within (D) Microvascular network integrated (red) within fibroblast containing dermis (green cells) showing capability to scale tissue size and characterize dynamic transport of therapeutics (blue nanoparticles) within the vessels (E) Tisssue-specific skin platform in which tissue properties and specific vascular patterns were generated based on clinical imaging data
In the fabrication of the multilayered skin model, differentiating keratinocytes were observed becoming corneocytes, which is characteristic of in vivo response of the human epidermis shown in Figure 1B. the skin model also includes a vascularized dermal skin model with fibroblasts (blue) with a confluent endothelialized blood vessel (red) (see Figure 1C), Further development of enabling the influence of blood perfusion.
The types of characterization employed include measurement of dynamic and spatial cell viability in its native state and in response to burn injury. As illustrated in Figure 2A, preliminary tissue injury following contact burns can be clearly distinguished with staining techniques. The dark region denotes dead cells and green cells are the live periphery where burn injury was sub-lethal. The cell-growth kinetics have been quantified in response to varying thermal conditions.
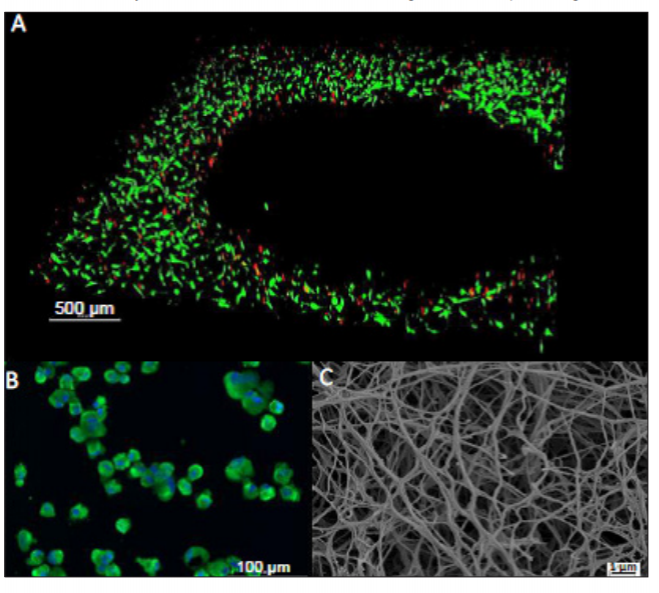
Figure 2: Characterization of the skin platform. (A) Burn injury profile in skin (green region is liver periphery and black central area is region of burn) (B) Heat shock protein expression of cells after burn (C) Scanning electron microscopy image of skin
Other types of cellular stress response markers, such as heat shock proteins, could serve as another metric of injury and a key parameter for predicting tissue survival and wound repair (see Figure 2B). The Rylander Group has developed the capability to determine mechanical properties of the skin platform determining how tissue integrity changes over time or in response to injury, which directly affects wound closure and healing. Structural properties of the skin platform determined using scanning electron microscopy shows tissue structure, porosity, and fiber diameter, and can be extended to any material characterization before and following thermal and chemical insults to understand the extent of material and tissue compromised (see Figure 2C).
The size of the tissue platform can be customized to desired dimensions and can mimic skin from different regions of the body. Researchers can scale the tissue size, tune the matrix and cellular composition, and incorporate multiple blood vessels or microvascular networks (see Figure 1D). The skin platform can also be adapted to represent patient or population-specific (e.g., gender, age, and ethnicity) types of skin by capturing tissue properties and vessel architecture from human imaging data (see Figure 1E). The platform can also remain stable at 37ºC for two weeks, allowing for long-termstudy and storage.
Recommendations for Further Research
Initially, the improved and validated burn simulants will be used principally in the way the current burn sensor injury models are used—implemented in a computational computer code that processes sensor data from flame and thermal testing of a textile/garment in order to predict the distribution and body surface areas that are theoretically burned as a result of the test exposure. These tissue phantoms may also be used in further development (as test devices in lieu of traditional instrumented forms) and as an experimental test bed to aid in the development of burn models for highly vasculated tissue.
As previously mentioned, the use of IEDs in OEF/OIF could be a contributing factor to the rise in combat-related burn injury rates. The burn patterns of IEDs typically affect the face and limbs [4], which rank first and second in burn injuries among the warfighters in Iraq and Afghanistan
[11]. This elevates the necessity of developing more accurate models that can be used to simulate these tissues. While the hands and face are not currently monitored in the standard ASTM F1930 manikin test, burns frequently occur in these tissues on the battlefield and have a significant impact on soldier quality of life. Blood perfusion through the human vasculature could strongly affect burn injury spread and severity by affecting physiologic burn response. To address fluid dynamics of burn injury, researchers are developing enhanced skin simulants, to include vasculature of veins/ capillary. In order to develop the most realistic models for testing clothing and gear used in the protection of these sensitive and vascular areas, more research should be done to characterize the locations and patterns of burns witnessed in veterans of OIF/OEF. As it appears that burn injury rates have not been updated since 2006 [1], additional research may be necessary to provide an accurate representation of the classification of burns and new rates.
The simulants may also be used as a testing mechanism to characterize, develop, and optimize current or future protective gear for Soldiers in-theater. The skin platform could provide information on cellular response to a wide range of insults (e.g., chemical, thermal) and in operational climates (e.g., desert, jungle). Furthermore, the simulant can also be observed on a molecular and/or cellular level to independently evaluate the severity of injury (by evaluating cell death and/or biomarkers, such as heat shock proteins) that may result from specific thermal (radiant, convective) threat situations, providing further insight into wound healing research. DoD operates in a broad range of environments in which the warfighter could be exposed to a variety of contaminants. It is impractical to conduct field research and be prepared for each of the environments. However, using phantoms to simulate multiple environments would allow DoD to conduct necessary research on how these different contaminants could affect the warfighter prior to combat, thus enabling commands to be equipped with the proper protective equipment.
Conclusion
The skin platform is a cost-effective, high-throughput system that can be used instead of animal skin or other stand-ins to provide a living, growing tissue that can be customized for any given research endeavor by optimizing its characteristics including size, shape, layer thickness, cell and protein composition, vasculature geometry, and heterogeneous features such as hair follicles and pores. This versatile research and development effort of tailored multi-layered skin platforms promises to be an interesting test bed, not only in Soldier protection, but for future medical research.
References
1. Kauvar, D. S., Wolf, S. E., Wade, C. E., Cancio, L. C., Renz, E. M., & Holcomb, J. B. (2006). Burns sustained in combat explosions in Operations Iraqi and Enduring Freedom (OIF/OEF explosion burns).
Burns, 32(7), 853-857. doi:10.1016/j.burns.2006.03.008
2. Escolas, S. M., Archuleta, D. J., Orman, J. A., Chung, K. K., & Renz, E. M. (2017). Postdischarge cause-of-death analysis of combat-related burn patients. Journal of Burn Care & Research, 38(1). doi:10.1097/
bcr.0000000000000319
3. Barzilai, L., Harats, M., Wiser, I., Weissman, O., Domniz, N., Glassberg, E., . . . Haik, J. (2015). Characteristics of improvised explosive device trauma casualties in the Gaza Strip and other combat regions: The Israeli experience [Abstract]. Wounds, 27(8). Retrieved from https://www.ncbi.nlm.nih.gov/pubmed/26284374
4. Cancio, L. C., Horvath, E. E., Barillo, D. J., Kopchinski, B. J., Charter, K. R., Montalvo,
A. E., . . . Holcomb, J. B. (2005). Burn support for Operation Iraqi Freedom and related operations, 2003 to 2004. Journal of Burn Care & Rehabilitation, 26(2), 151-161.doi:10.1097/01.bcr.0000155540.31879.fb
5. Champion, H. R., Bellamy, R. F., Roberts, C. P., & Leppaniemi, A. (2003). A profile of combat injury. Journal of Trauma, 54(5), S13-S19. doi:10.1097/01. TA.0000057151.02906.27
6. International Organization for Standardization. (2003). ISO 17492:2003, Clothing for protection against heat and flame — Determination of heat transmission on exposure to both flame and radiant heat. Retrieved from https://www.iso.org/standard/30698.html
7. ASTM International. (2017). ASTM F1930-17, Standard Test Method for Evaluation of Flame Resistant Clothing for Protection Against Fire Simulations Using an Instrumented Manikin. West Conshohocken, PA: ASTM International. Retrieved from https://www.astm.org/Standards/F1930.htm
8. Stoll, A. M., & Greene, L. C. (1959). Relationship between pain and tissue damage due to thermal radiation. Journal of Applied Physiology, 14(3), 373-382.
9. Stoll, A. M., & Chianta, M. A. (1971). Heat transfer through fabrics as related to thermal injury. Transactions of the New York Academy of Sciences, 33(7 Series II), 649-670. doi:10.1111/j.2164-0947.1971.tb02630.x
10. Stoll, A. M., & Chianta, M. A. (1968). Burn production and prevention in convective and radiant heat transfer. Aerospace Medicine, 39(10), 1097-1100.
11. Wolf, S. E., Kauvar, D. S., Wade, C. E., Cancio, L. C., Renz, E. P., Horvath, E. E., . . . Holcomb, J. B. (2006). Comparison Between Civilian Burns and Combat Burns From Operation Iraqi Freedom and Operation Enduring Freedom. Annals of Surgery, 243(6), 786-795. doi:10.1097/01.sla.0000219645.88867.b7


