Much of the dietary information provided to our military personnel is modeled after outdated recommendations that emphasize processed carbohydrates and sugary sports drinks [1]. Unlike fat and protein, there is no obligate need for carbohydrate consumption in humans [2]. It is well established that organs and tissues in the body readily adapt to fatty acid and ketone fueling [3]. Accumulating evidence indicates that increased availability and utilization of these lipid-based fuels is associated with favorable metabolic and physiologic outcomes, including reduction of fat mass relative to lean mass, enhancement of physical and cognitive measures during aging, and improvement of cardiac performance in response to physical or environmental stress. A well-formulated ketogenic diet (KD) that emphasizes carbohydrate restriction, moderate-protein, fat to satiety, and nutritional ketosis, represents a credible method of supporting physical performance and improving health.
Low Carbohydrate, Ketogenic Diets, and Ketone Metabolism
KDs deviate from traditional high-carbohydrate diets, as well as differing from many popular low-carbohydrate approaches (e.g., Paleo diets) that do not elevate ketones. A well-formulated KD includes limited carbohydrates (typically 50g/day or less) and adequate protein (1.2-2.0 g/kg Reference Weight). The remaining calories are derived from fat, which is consumed to satiety (~70-80 percent total kcal/ day on a eucaloric weight-maintenance KD) (see Figure 1).
KDs result in reduced availability of glucose and stimulation of insulin. This metabolic and hormonal state stimulates adipose tissue lipolysis and enhanced fatty acid delivery to tissues that manifests in profound increases in fatty acid oxidation and hepatic ketogenesis.
Ketogenesis and associated metabolic machinery are engrained in human biology as it was necessary to the survival of our ancestors [7]. Following an overnight fast, blood ketone concentrations may reach 0.1-0.3 mM. KDs typically result in ketone concentration between 0.5 mM to 3mM (i.e., nutritional ketosis). The two primary circulating ketones are Beta Hydroxybutyrate (βHB) and acetoacetate. Physiological elevation of blood ketones via KD [8-11] is safe and ensures adequate fuel supply to all organs during periods of decreased glucose availability.
Ketosis is not analogous with ketoacidosis, which is a life-threatening condition typified by hyperglycemia (BG>250 mg/dL), blood acidosis (pH10Mm) hyperketonemia [12, 13]. Risk of ketoacidosis is a concern in uncontrolled type 1 diabetics. Conversely, individuals who produce insulin possess an exquisite feedback mechanism in which βHB provides direct negative feedback inhibition of adipocyte lipolysis, as well as indirect stimulation of insulin secretion—thus preventing ketoacidosis risk [14].

Figure 1. Macronutrient distribution associated with differing dietary paradigms. Based upon 3200-4200 kcal/day. Approximate energy intake needs reported previously [11-13].
Addressing Current Challenges Through a KD Nutrition Strategy
Since the early 1900s, LCDs and KDs have been implemented and studied in neurological diseases [15, 16], weight loss [17-19, 9], insulin resistance [9, 20], cardio-metabolic disease [17, 18, 21, 22], and cancer [23, 24]. Evidence in support of implementing a KD as a therapy to address various pathophysiological and chronic disease challenges in human health [8, 22, 10, 11] has increased dramatically in the past 10-15 years. Healthy, untrained individuals, patients, and athletes are increasingly aware of research related to KDs. As such, KDs should not be dismissed as a fringe concept or fad diet.
A significant body of work indicates that “nutritional” or “physiological” ketosis (defined as circulating ketones between 0.5-5.0 mM) has clinical [25, 26] and performance [27-30] applications due to potent metabolic and signaling changes in humans [31-37]. Additionally, there are many military-relevant health and performance challenges that may be affected by nutritional ketosis, as can be seen in Figure 2.
Body Composition, Overweight, Obesity Rates
High rates of overweight and obesity present obstacles in military recruitment and physical readiness. Over two thirds (~71-75 percent of Americans within military age range (~17- 24 years) are classified as unfit to enlist [38, 39]. This issue has been cited as a “national security concern” for DoD recruitment and homeland defense at large [40]. Throughout the military, food selection is being investigated in an effort to improve dietary intake amongst military personnel [41]. Body composition improvements (reductions in fat mass relative to lean mass) are amongst the most consistent outcomes reported with KD interventions [42, 32, 43, 33, 44-46, 31].
Several published primary research studies [22, 19], reviews [17], and meta-analysis [47] conclude that a KD is superior to a low fat diet in promoting weight loss without explicit instruction to reduce calories.
Disease Protection & Longevity: Non-metabolic roles of Ketones
In addition to serving as a preferred fuel, βHB is a potent signaling molecule [48] capable of regulating aspects of gene expression, inflammation, oxidative stress, and longevity [48, 49]. Data published in Science in 2013 [50] demonstrated that βHB inhibits class I histone deacetylases (HDAC). HDAC inhibition enhances expression of several genes which increase antioxidant production. βHB has also been shown to inhibit the NLRP3 inflammasome [51], which is recognized as an important mediator of inflammation and metabolic dysfunction related to obesity.
In 2017, two separate groups published provocative evidence that ketosis enhanced “healthspan” with improved physical and cognitive measures during aging [48, 49]. Additionally, high density lipoprotein and triglyceride concentrations, both markers for risk of heart disease and stroke, are consistently improved with KDs [47]. Lack of data in defense health reports indicates further research is needed to explore enhanced resilience, oxidative protection, and healthspan benefits associated with ketosis in military personnel.
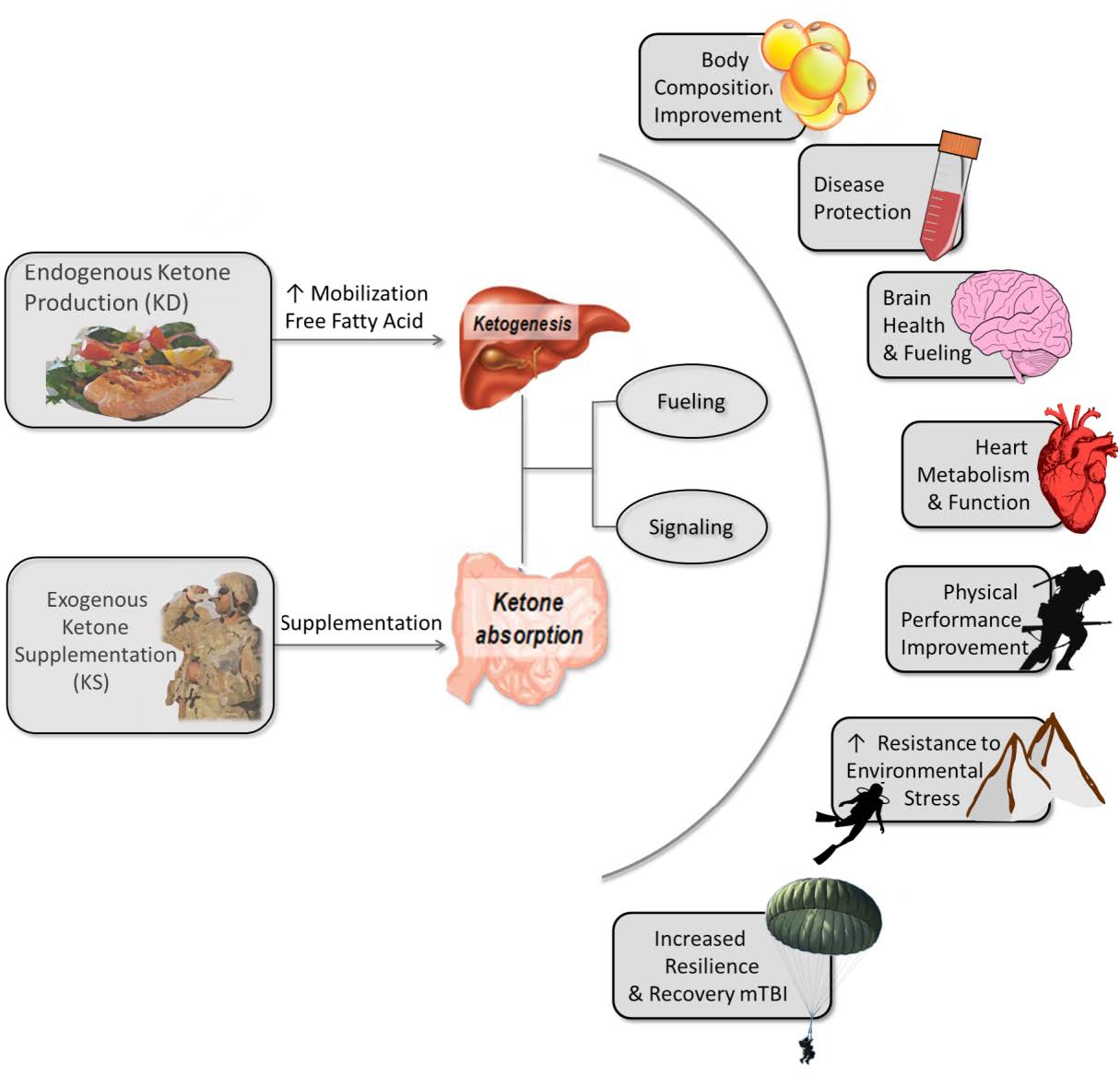
Figure 2. Physiological elevation of ketones in humans may have military relevant effects on health and performance
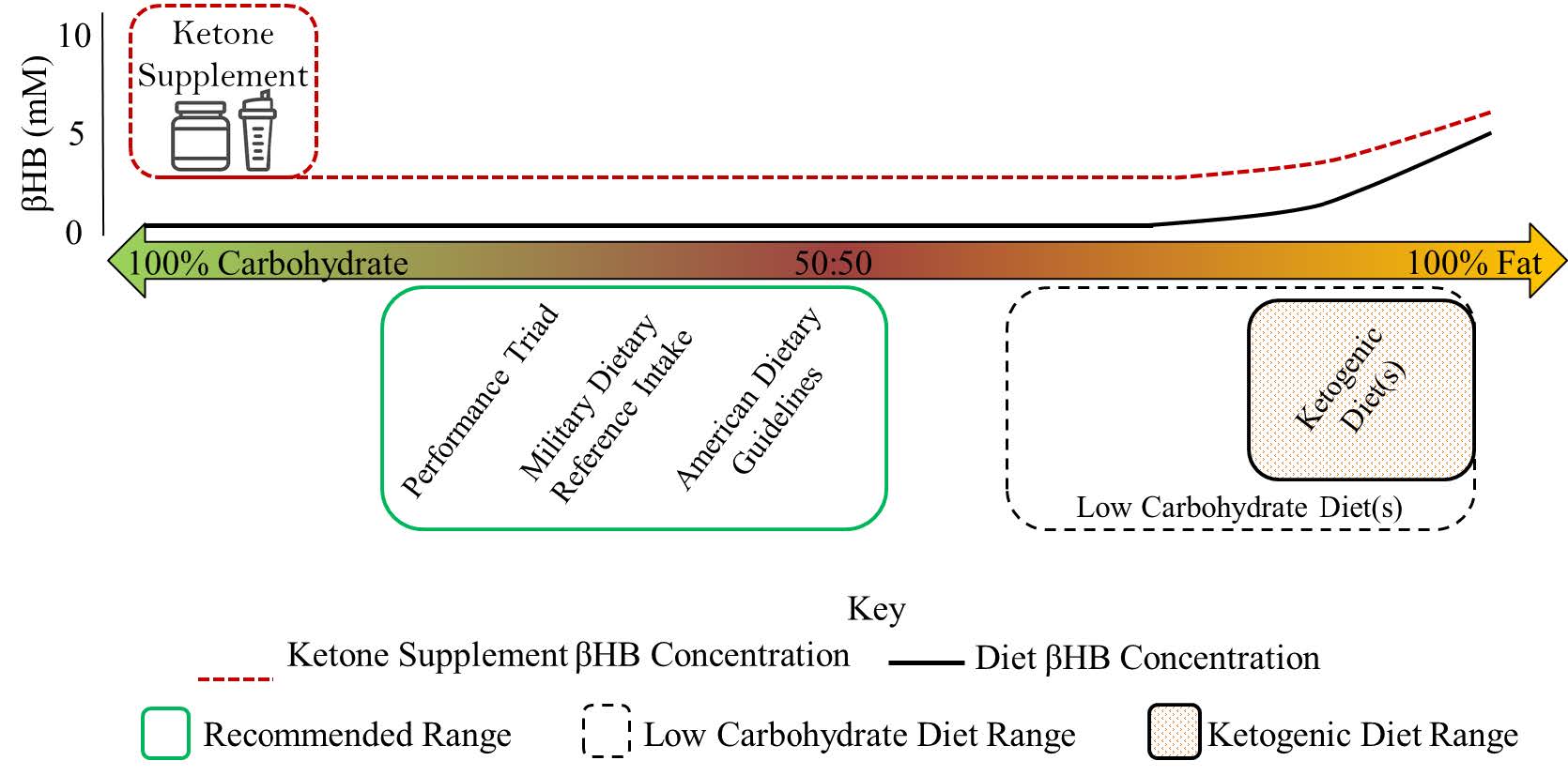
Figure 3. βHB concentration related to dietary carbohydrate vs. fat macronutrient intake and exogenous ketone supplementation
Brain Health & Central Nervous System Fueling
The brain preferentially uses ketones as a stable and sustainable fuel source. Clinically, KDs were introduced a century ago as an effective treatment to decrease seizures in patients with epilepsy [52-56]. Since then, researchers have produced encouraging evidence for KD use in the prevention and treatment of neurological diseases in addition to epilepsy, such as Alzheimers [16].
According to the results of recent studies, KDs could potentially improve the health and performance of military divers. In a small study of three trained high pressure divers, short duration KDs were shown to lengthen dive time and prevent complications associated with CNS toxicity in divers [57].
Researchers recommended KDs, lasting periods as short as 48 hours, as a tool to improve safety and dive performance. Additionally, results from a separate diver study suggest that a KD may protect against lipid peroxidation and inflammation caused by high pressure enriched air nitrox diving [58].
Military personnel struggling with sleep loss [59], traumatic brain injury [60], and post-traumatic stress disorder or depression [61, 62] may experience mood stabilization in part due to elevated blood ketone levels [63]. There is further evidence that ketones may support recovery from brain injury [64, 65]. Primary research and reviews demonstrate cognitive health and performance benefits associated with ketosis [66, 67, 26, 34, 68].
Human research in tactical groups is needed to establish the direct translation and downstream effects on safety, operational success, as well as precision in training scenarios and combat operations. There are many promising lines of evidence [69, 70] that show improved CNS function with elevated ketone levels based primarily on animal studies [71, 72] and human trials in epilepsy [73], Alzheimer’s [34], and Parkinson’s [74] patients. Ketosis and brain health is one of the most important areas for further study likely to yield significant returns on investment.
Heart Metabolism and Function
Like the brain, the heart has high energy requirements (≥300 kcal/day) and displays impressive metabolic flexibility. The heart readily utilizes ketones in direct proportion to plasma concentration [75]. Increased ketone utilization may positively impact heart function and improve heart efficiency [54, 76]. Cardiac performance is critical in response to physical, environmental, or disease stress [77]. Ketone elevations may provide unique performance advantages to warfighters and tactical athletes in multi-stressor combat scenarios or otherwise. Although in-vivo data are lacking, multiple findings from as recently as 2016 demonstrate that an extended duration KD is a promising strategy for improved heart function in healthy individuals, and may mitigate metabolic dysregulation and consequent functional decline associated with disease states of the myocardium [78, 26].
Ketone fueling has been recommended as potential nutritional enhancement for the warfighter to enhance physical fitness as well as to prevent “avoidable maladies” such as cardiovascular disease [79, 22]. Physiological ketosis is worth investigating as a strategy to optimize cardiac metabolism and function in healthy individuals exposed to external stressors in the battlefield—specifically altitude [80, 81], which exerts additional strain on the cardiopulmonary system during combat scenarios.
Physical Performance & Environmental Stress Resistance
In nutritional ketosis, the metabolic needs of organs/tissues are met primarily by ketones and fatty acids. Endogenous fuels storage of fat is, minimally, an order of magnitude larger than carbohydrate storage supply. In various combat scenarios, calorie intake is often inadequate [6] or fasting may be required for >12-24 hours. Keto-adapted individuals would have a significant advantage during these situations in that organs remain well-nourished offering resistance to hypoglycemia induced degradation in performance and cognition.
Strenuous exertion during hypoglycemia can result in syncope [82, 83], thus compromising safety and mission effectiveness. In addition to dramatically increased fat oxidation rates (>1.5g/min), skeletal muscle biopsy data from elite trained endurance athletes indicates there are minimal effects on storage, utilization, and re-synthesis of skeletal muscle glycogen in keto-adapted individuals [27].
Coupled with reductions in fat mass, increased fat metabolism [36, 27, 32], relative power output [31], and stamina improvement [37], power to weight ratio enhancement is expected in those that adapt to a KD [29, 84]. Applied across a range of Military Occupational Specialties, this may be an important consideration as non-combat injury rates remain prevalent [85, 86]. In addition to performance benefits, KDs may address injury concerns in two ways. First, leaner soldiers endure less musculoskeletal injuries [87].
Second, KDs alter fueling such that fatigue effects are minimized based upon time to exhaustion performance tests [35, 37]. Leaner soldiers, less affected by fatigue, would have attenuated injury risk which would translate into desirable downstream effects on overall soldier health [88], duty time effectiveness [89], and financial burden [90].
Deploying Ketosis: An Ordinance with Large Scale Health and Performance Potential
Nutrition is shifting based on accumulating evidence that shows the dietary paradigms such as a KD have therapeutic and health/performance-enhancing effects. There is growing interest in ketones as evidenced by the metabolic dominance program put forth by the Defense Advanced Research Projects Agency [91], which supported work to develop exogenous ketone supplements (KS). It is unclear if KS, which may induce greater elevation in blood ketones than KDs (see Figure 3), produce similar fueling and signaling benefits of KDs outlined in this article.
Strategies which leverage ketone fuels may be advantageous in addressing a range of challenges affecting military [92] and civilian populations [93]. Enhanced fueling and signaling from ketones may provide tactical advantages in personnel exposed to militarily unique scenarios involving physical, cognitive, and environmental stressors [79]. Research evidence has reached a critical mass in support of further investigation of KDs and/or KS strategies in relation to human health and tactical performance across diverse Military Occupational Specialties.
References
- MEDCOM. (2015). Triad: P3 Soldier Guide. San Antonio: U.S. Army Medical Command.
- Westman, E. (2002). Is dietary carbohydrate essential for human nutrition?. The American Journal Of Clinical Nutrition, 75(5), 951- 953. doi: 10.1093/ajcn/75.5.951a
- Cahill G., Jr., Felig P., Owen O., Wahren J. (1970). Metabolic adaptation to prolonged starvation in man. Nord Med, 83(3), 89.
- Margolis, L., Crombie, A., McClung, H., McGraw, S., Rood, J., Montain, S., & Young, A. (2014). Energy Requirements of US Army Special Operation Forces During Military Training. Nutrients, 6(5), 1945-1955. doi: 10.3390/nu6051945
- Tharion, W., Lieberman, H., Montain, S., Young, A., Baker-Fulco, C., DeLany, J., & Hoyt, R. (2005). Energy requirements of military personnel. Appetite, 44(1), 47-65. doi: 10.1016/j.appet.2003.11.010
- Carlson-Newberry, S., Costello, R., & Poos, M. (1999). Committee on Military Nutrition Research. Washington, D.C.: National Academy Press.
- Newman, J., & Verdin, E. (2014). Ketone bodies as signaling metabolites. Trends In Endocrinology & Metabolism, 25(1), 42-52. doi: 10.1016/j.tem.2013.09.002
- Feinman, R., Pogozelski, W., Astrup, A., Bernstein, R., Fine, E., Westman, E. … & Worm, N. (2015). Dietary carbohydrate restriction as the first approach in diabetes management: Critical review and evidence base. Nutrition, 31(1), 1-13. doi: 10.1016/j. nut.2014.06.011
- McKenzie, A., Hallberg, S., Creighton, B., Volk, B., Link, T., Abner, M. … & Phinney, S. (2017). A Novel Intervention Including Individualized Nutritional Recommendations Reduces Hemoglobin A1c Level, Medication Use, and Weight in Type 2 Diabetes. JMIR Diabetes, 2(1), e5. doi: 10.2196/diabetes.6981
- Paoli, A., Rubini, A., Volek, J., & Grimaldi, K. (2013). Beyond weight loss: a review of the therapeutic uses of very-low-carbohydrate (ketogenic) diets. European Journal Of Clinical Nutrition, 67(8), 789-796. doi: 10.1038/ ejcn.2013.116
- Paoli A., Canato M., Toniolo L., Bargossi A.M., Neri M., Mediati M., … & Bianco, A. (2011) . The ketogenic diet: an underappreciated therapeutic option? Clin Ter, 162(5), e145-153.
- Naunheim, R., Jang, T., Banet, G., Richmond, A., & McGill, J. (2006). Point-of-care Test Identifies Diabetic Ketoacidosis at Triage. Academic Emergency Medicine, 13(6), 683-685. doi: 10.1197/j.aem.2006.01.020
- Umpierrez, G., DiGirolamo, M., Tuvlin, J., Isaacs, S., Bhoola, S., & Kokko, J. (2000). Differences in metabolic and hormonal milieu in diabetic- and alcohol-induced ketoacidosis. Journal Of Critical Care, 15(2), 52-59. doi: 10.1053/jcrc.2000.7900
- Robinson, A., & Williamson, D. (1980). Physiological roles of ketone bodies as substrates and signals in mammalian tissues. Physiological Reviews, 60(1), 143-187. doi: 10.1152/physrev.1980.60.1.143
- Cunnane, S., Nugent, S., Roy, M., Courchesne-Loyer, A., Croteau, E., & Tremblay, S., … & Rapoport, S. (2011). Brain fuel metabolism, aging, and Alzheimer’s disease. Nutrition, 27(1), 3-20. doi: 10.1016/j. nut.2010.07.021
- Stafstrom, C., & Rho, J. (2012). The Ketogenic Diet as a Treatment Paradigm for Diverse Neurological Disorders. Frontiers In Pharmacology, 3. doi: 10.3389/ fphar.2012.00059
- Bravata, D., Sanders, L., Huang, J., Krumholz, H., Olkin, I., Gardner, C., & Bravata, D. (2003). Efficacy and Safety of Low-Carbohydrate Diets. JAMA, 289(14), 1837. doi: 10.1001/jama.289.14.1837
- Stern, L., Iqbal, N., Seshadri, P., Chicano, K., Daily, D., & McGrory, J., … & Samaha, F. (2004). The Effects of Low-Carbohydrate versus Conventional Weight Loss Diets in Severely Obese Adults: One-Year Follow-up of a Randomized Trial. Annals Of Internal Medicine, 140(10), 778. doi: 10.7326/0003- 4819-140-10-200405180-00007
- Volek, J., Sharman, M., Gómez, A., Judelson, D., Rubin, M., & Watson, G., … & Kraemer, W. (2004). Comparison of energy-restricted very low-carbohydrate and low-fat diets on weight loss and body composition in overweight men and women. Nutrition & Metabolism, 1(1), 13. doi: 10.1186/1743-7075-1-13
- Hallberg, S., McKenzie, A., Williams, P., Bhanpuri, N., Peters, A., & Campbell, W., … & Volek, J. (2018). Effectiveness and Safety of a Novel Care Model for the Management of Type 2 Diabetes at 1 Year: An Open-Label, Non-Randomized, Controlled Study. Diabetes Therapy, 9(2), 583-612. doi: 10.1007/s13300-018-0373-9
- Bazzano, L., Hu, T., Reynolds, K., Yao, L., Bunol, C., & Liu, Y., … & He, J. (2014). Effects of Low-Carbohydrate and Low-Fat Diets. Annals Of Internal Medicine, 161(5), 309. doi: 10.7326/m14-0180
- Sharman, M., Kraemer, W., Love, D., Avery, N., Gómez, A., Scheett, T., & Volek, J. (2002). A Ketogenic Diet Favorably Affects Serum Biomarkers for Cardiovascular Disease in Normal-Weight Men. The Journal Of Nutrition, 132(7), 1879-1885. doi: 10.1093/ jn/132.7.1879
- Masino, S. (2016). Ketogenic diet and metabolic therapies. New York: Oxford University Press.
- Hyde, P., Lustberg, M., Miller, V., LaFountain, R., & Volek, J. (2017). Pleiotropic effects of nutritional ketosis: Conceptual framework for keto-adaptation as a breast cancer therapy. Cancer Treatment And Research Communications, 12, 32-39. doi: 10.1016/j.ctarc.2017.06.001
- Volek, J., Phinney, S., Kossoff, E., Eberstein, J., & Moore, J. (2011). The art and science of low carbohydrate living. Lexington, KY: Beyond Obesity.
- Veech, R. (2004). The therapeutic implications of ketone bodies: the effects of ketone bodies in pathological conditions: ketosis, ketogenic diet, redox states, insulin resistance, and mitochondrial metabolism. Prostaglandins, Leukotrienes And Essential Fatty Acids, 70(3), 309-319. doi: 10.1016/j. plefa.2003.09.007
- Volek, J., Freidenreich, D., Saenz, C., Kunces, L., Creighton, B., & Bartley, J., … & Phinney, S. (2016). Metabolic characteristics of keto-adapted ultra-endurance runners. Metabolism, 65(3), 100-110. doi: 10.1016/j.metabol.2015.10.028
- Volek, J., Noakes, T., & Phinney, S. (2014). Rethinking fat as a fuel for endurance exercise. European Journal Of Sport Science, 15(1), 13-20. doi: 10.1080/17461391.2014.959564
- Noakes, T., Volek, J., & Phinney, S. (2014). Low-carbohydrate diets for athletes: what evidence?. British Journal Of Sports Medicine, 48(14), 1077-1078. doi: 10.1136/ bjsports-2014-093824
- Paoli, A., Bianco, A., & Grimaldi, K. (2015). The Ketogenic Diet and Sport. Exercise And Sport Sciences Reviews, 43(3), 153-162. doi: 10.1249/jes.0000000000000050
- McSwiney, F., Wardrop, B., Hyde, P., Lafountain, R., Volek, J., & Doyle, L. (2018). Keto-adaptation enhances exercise performance and body composition responses to training in endurance athletes. Metabolism, 81, 25-34. doi: 10.1016/j.metabol.2017.10.010
- Zinn, C., Wood, M., Williden, M., Chatterton, S., & Maunder, E. (2017). Ketogenic diet benefits body composition and well-being but not performance in a pilot case study of New Zealand endurance athletes. Journal Of The International Society Of Sports Nutrition, 14(1), 22. doi: 10.1186/s12970- 017-0180-0
- Vargas, S., Romance, R., Petro, J., Bonilla, D., Galancho, I., & Espinar, S., … & Benitez-Porres,J. (2018). Efficacy of ketogenic diet on body composition during resistance training in trained men: a randomized controlled trial. Journal Of The International Society Of Sports Nutrition, 15(1), 31. doi: 10.1186/s12970-018-0236-9
- Cunnane, S., Courchesne-Loyer, A., StPierre, V., Vandenberghe, C., Pierotti, T., & Fortier, M, … & Castellano, C. (2016). Can ketones compensate for deteriorating brain glucose uptake during aging? Implications for the risk and treatment of Alzheimer’s disease. Annals Of The New York Academy Of Sciences, 1367(1), 12-20. doi: 10.1111/ nyas.12999
- Phinney, S., Bistrian, B., Evans, W., Gervino, E., & Blackburn, G. (1983). The human metabolic response to chronic ketosis without caloric restriction: Preservation of submaximal exercise capability with reduced carbohydrate oxidation. Metabolism, 32(8), 769-776. doi: 10.1016/0026- 0495(83)90106-3
- Phinney, S., Bistrian, B., Wolfe, R., & Blackburn, G. (1983). The human metabolic response to chronic ketosis without caloric restriction: Physical and biochemical adaptation. Metabolism, 32(8), 757-768. doi: 10.1016/0026-0495(83)90105-1
- Phinney, S., Horton, E., Sims, E., Hanson, J., Danforth, E., & Lagrange, B. (1980). Capacity for Moderate Exercise in Obese Subjects after Adaptation to a Hypocaloric, Ketogenic Diet. Journal Of Clinical Investigation, 66(5), 1152-1161. doi: 10.1172/ jci109945
- Cawley, J., & Maclean, J. (2011). UNFIT FOR SERVICE: THE IMPLICATIONS OF RISING OBESITY FOR US MILITARY RECRUITMENT. Health Economics, 21(11), 1348-1366. doi: 10.1002/hec.1794
- Christeson W., Taggart A., Messner-Zidell S. (2009) Ready, Willing, and Unable to Serve: 75 Percent of Young Adults Cannot Join the Military: Early Education Across America is Needed to Ensure National Security. Retrieved from http://cdn.missionreadiness.org/NATEE1109.pdf
- Davis, K. (2011). Obesity: A National Security Concern. Carlisle: U.S. Army War College.
- Harkins, G. (2018). The Military Is Overhauling Troops’ Chow as Obesity Rates Soar. Retrieved from https://www.military.com/daily-news/2018/08/19/military-overhauling-troops-chow-obesity-rates-soar.html
- Jabekk, P., Moe, I., Meen, H., Tomten, S., & Høstmark, A. (2010). Resistance training in overweight women on a ketogenic diet conserved lean body mass while reducing body fat. Nutrition & Metabolism, 7(1), 17. doi: 10.1186/1743-7075-7-17
- Tinsley, G., & Willoughby, D. (2016). FatFree Mass Changes during Ketogenic Diets and the Potential Role of Resistance Training. International Journal Of Sport Nutrition And Exercise Metabolism, 26(1), 78-92. doi: 10.1123/ijsnem.2015-0070
- Wilson, J., Lowery, R., Roberts, M., Sharp, M., Joy, J., & Shields, K., … & D’Agostino, D. (2017). The Effects of Ketogenic Dieting on Body Composition, Strength, Power, and Hormonal Profiles in Resistance Training Males. Journal Of Strength And Conditioning Research, 1. doi: 10.1519/ jsc.0000000000001935
- Zajac, A., Poprzecki, S., Maszczyk, A., Czuba, M., Michalczyk, M., & Zydek, G. (2014). The Effects of a Ketogenic Diet on Exercise Metabolism and Physical Performance in Off-Road Cyclists. Nutrients, 6(7), 2493- 2508. doi: 10.3390/nu6072493
- Rhyu, H., & Cho, S. (2014). The effect of weight loss by ketogenic diet on the body composition, performance-related physical fitness factors and cytokines of Taekwondo athletes. Journal Of Exercise Rehabilitation, 10(5), 326-331. doi: 10.12965/jer.140160
- Sackner-Bernstein, J., Kanter, D., & Kaul, S. (2015). Dietary Intervention for Overweight and Obese Adults: Comparison of Low-Carbohydrate and Low-Fat Diets. A Meta-Analysis. PLOS ONE, 10(10), e0139817. doi: 10.1371/journal.pone.0139817
- Newman, J., Covarrubias, A., Zhao, M., Yu, X., Gut, P., & Ng, C., … & Verdin, E. (2017). Ketogenic Diet Reduces Midlife Mortality and Improves Memory in Aging Mice. Cell Metabolism, 26(3), 547-557.e8. doi: 10.1016/j.cmet.2017.08.004
- Roberts, M., Wallace, M., Tomilov, A., Zhou, Z., Marcotte, G., & Tran, D., … & Lopez-Dominguez, J. (2017). A Ketogenic Diet Extends Longevity and Healthspan in Adult Mice. Cell Metabolism, 26(3), 539-546.e5. doi: 10.1016/j.cmet.2017.08.005
- Shimazu, T., Hirschey, M., Newman, J., He, W., Shirakawa, K., & Le Moan, N., … & Verdin, E. (2012). Suppression of Oxidative Stress by -Hydroxybutyrate, an Endogenous Histone Deacetylase Inhibitor. Science, 339(6116), 211-214. doi: 10.1126/ science.1227166
- Youm, Y., Nguyen, K., Grant, R., Goldberg, E., Bodogai, M., & Kim, D., … & Dixit, V. (2015). The ketone metabolite β-hydroxybutyrate blocks NLRP3 inflammasome– mediated inflammatory disease. Nature Medicine, 21(3), 263-269. doi: 10.1038/ nm.3804
- Gaby, A.R. (2007). Natural approaches to epilepsy. Alternative medicine review : a journal of clinical therapeutic, 12(1), 9-24
- Kossoff, E. (2004). More fat and fewer seizures: dietary therapies for epilepsy. The Lancet Neurology, 3(7), 415-420. doi: 10.1016/s1474-4422(04)00807-5
- L. Veech, Britton Chance, Yoshihiro, R. (2001). Ketone Bodies, Potential Therapeutic Uses. IUBMB Life (International Union Of Biochemistry And Molecular Biology: Life), 51(4), 241-247. doi: 10.1080/152165401753311780
- Wiznitzer, M. (2008). From observations to trials: the ketogenic diet and epilepsy. The Lancet Neurology, 7(6), 471-472. doi: 10.1016/s1474-4422(08)70093-0
- Wilder, R. (1921). The effects of ketonemia on the course of epilepsy. Mayo Clin Proc, 2, 307-308
- Valadao, J., Vigilante, J., DiGeorge, N., O’Connor, S., Bear, A., Kenyon, J., … & H. Whelan. (2014). Ketogenic diet for high partial pressure oxygen diving. Undersea & Hyperbaric Medicine, 41(4), 331-335.
- Bosco, G., Rizzato, A., Quartesan, S., Camporesi, E., Mangar, D., & Paganini, M., … & Paoli, M. (2018). Effects of the Ketogenic diet in overweight divers breathing Enriched Air Nitrox. Scientific Reports, 8(1). doi: 10.1038/s41598-018-20933-w
- A. Caldwell, J., Knapik, J., & Lieberman, H. (2017). Trends and factors associated with insomnia and sleep apnea in all United States military service members from 2005 to 2014. Journal Of Sleep Research, 26(5), 665-670. doi: 10.1111/jsr.12543
- Wojcik, B., Stein, C., Bagg, K., Humphrey, R., & Orosco, J. (2010). Traumatic Brain Injury Hospitalizations of U.S. Army Soldiers Deployed to Afghanistan and Iraq. American Journal Of Preventive Medicine, 38(1), S108-S116. doi: 10.1016/j.amepre.2009.10.006
- Murphy, P., & Burnham, W. (2006). The ketogenic diet causes a reversible decrease in activity level in Long–Evans rats. Experimental Neurology, 201(1), 84-89. doi: 10.1016/j.expneurol.2006.03.024
- Murphy, P., Likhodii, S., Nylen, K., & Burnham, W. (2004). The antidepressant properties of the ketogenic diet. Biological Psychiatry, 56(12), 981-983. doi: 10.1016/j. biopsych.2004.09.019
- El-Mallakh, R., & Paskitti, M. (2001). The ketogenic diet may have mood-stabilizing properties. Medical Hypotheses, 57(6), 724- 726. doi: 10.1054/mehy.2001.1446
- White, H., & Venkatesh, B. (2011). Clinical review: Ketones and brain injury. Critical Care, 15(2), 219. doi: 10.1186/cc10020
- Prins, M., & Matsumoto, J. (2014). The collective therapeutic potential of cerebral ketone metabolism in traumatic brain injury. Journal Of Lipid Research, 55(12), 2450- 2457. doi: 10.1194/jlr.r046706
- Krikorian, R., Shidler, M., Dangelo, K., Couch, S., Benoit, S., & Clegg, D. (2012). Dietary ketosis enhances memory in mild cognitive impairment. Neurobiology Of Aging, 33(2), 425.e19-425.e27. doi: 10.1016/j. neurobiolaging.2010.10.006
- Evans, M., Cogan, K., & Egan, B. (2016). Metabolism of ketone bodies during exercise and training: physiological basis for exogenous supplementation. The Journal Of Physiology, 595(9), 2857-2871. doi: 10.1113/jp273185
- Hallböök, T., Ji, S., Maudsley, S., & Martin, B. (2012). The effects of the ketogenic diet on behavior and cognition. Epilepsy Research, 100(3), 304-309. doi: 10.1016/j. eplepsyres.2011.04.017
- Gasior, M., Rogawski, M. A., & Hartman, A. L. (2006). Neuroprotective and disease-modifying effects of the ketogenic diet. Behavioural Pharmacology, 17(5-6), 431-439.
- Ruskin, D., & Masino, S. (2012). The Nervous System and Metabolic Dysregulation: Emerging Evidence Converges on Ketogenic Diet Therapy. Frontiers In Neuroscience, 6(33). doi: 10.3389/fnins.2012.00033
- Balietti, M., Giorgetti, B., Di Stefano, G., Casoli, T., Platano, D., & Solazzi, M., … & Fattoretti, P. (2010). A ketogenic diet increases succinic dehydrogenase (SDH) activity and recovers age-related decrease in numeric density of SDH-positive mitochondria in cerebellar Purkinje cells of late-adult rats. Micron, 41(2), 143-148. doi: 10.1016/j. micron.2009.08.010
- Kashiwaya, Y., Bergman, C., Lee, J., Wan, R., King, M., & Mughal, M., … & Veech, R. (2013). A ketone ester diet exhibits anxiolytic and cognition-sparing properties, and lessens amyloid and tau pathologies in a mouse model of Alzheimer’s disease. Neurobiology Of Aging, 34(6), 1530-1539. doi: 10.1016/j.neurobiolaging.2012.11.023
- Freeman, J., Vining, E., Pillas, D., Pyzik, P., Casey, J., & Kelly, L. (1998). The Efficacy of the Ketogenic Diet-1998: A Prospective Evaluation of Intervention in 150 Children. PEDIATRICS, 102(6), 1358-1363. doi: 10.1542/peds.102.6.1358
- VanItallie, T., Nonas, C., Di Rocco, A., Boyar, K., Hyams, K., & Heymsfield, S. (2005). Treatment of Parkinson disease with diet-induced hyperketonemia: A feasibility study. Neurology, 64(4), 728-730. doi: 10.1212/01. wnl.0000152046.11390.45
- Bing, R., Siegel, A., Ungar, I., & Gilbert, M. (1954). Metabolism of the human heart: II. Studies on fat, ketone and acid metabolism. The American Journal Of Medicine, 16(4), 504-515. doi: 10.1016/0002-9343(54)90365- 4
- Sato, K., Kashiwaya, Y., Keon, C., Tsuchiya, N., King, M., Radda, G., … & Veech, R. L. (1995). Insulin, ketone bodies, and mitochondrial energy transduction. The FASEB Journal, 9(8), 651-658.
- A Aubert, G., Martin, O., Horton, J., Lai, L., Vega, R., & Leone, T., … & Kelly, D. (2016). The Failing Heart Relies on Ketone Bodies as a Fuel. Circulation, 133(8), 698-705. doi: 10.1161/circulationaha.115.017355
- Ferrannini, E., Mark, M., & Mayoux, E. (2016). CV Protection in the EMPA-REG OUTCOME Trial: A “Thrifty Substrate” Hypothesis. Diabetes Care, 39(7), 1108-1114. doi: 10.2337/dc16-0330
- Ford, K., & Glymour, C. (2014). The enhanced warfighter. Bulletin Of The Atomic Scientists, 70(1), 43-53. doi: 10.1177/0096340213516746
- Grau, L., & Jorgensen, W. (2003). Medical implications of high altitude combat. FOREIGN MILITARY STUDIES OFFICE (ARMY) FORT LEAVENWORTH KS.
- Acosta, M. (2003). High altitude warfare: The Kargil conflict and the future. NAVAL POSTGRADUATE SCHOOL MONTEREY CA.
- Gordon, B., Kohn, L., Levine, S., Matton, M., Scriver, W., & Whiting, W. (1925). Sugar Content Of The Blood In Runners Following A Marathon Race. Journal Of The American Medical Association, 85(7), 508. doi: 10.1001/jama.1925.02670070028009
- Levine, S., Gordon, B., & Derick, C. (1924). Some Changes In The Chemical Constituents Of The Blood Following A Marathon Race. Journal Of The American Medical Association, 82(22), 1778. doi: 10.1001/ jama.1924.02650480034015
- Phinney, S. (2004). Ketogenic diets and physical performance. Nutrition & Metabolism, 1(1), 2. doi: 10.1186/1743-7075-1-2
- Sell, T., Abt, J., Crawford, K., Lovalekar, M., Nagai, T., Deluzio, J., … & Lephart, S. (2010). Warrior Model for Human Performance and Injury Prevention: Eagle Tactical Athlete Program (ETAP) Part I. Journal of special operations medicine : a peer reviewed journal for SOF medical professionals, 10(4), 2-21
- Le, T., Gurney, J., Nnamani, N., Gross, K., Chung, K., & Stockinger, Z., … & Akers, K. (2018). A 12-Year Analysis of Nonbattle Injury Among US Service Members Deployed to Iraq and Afghanistan. JAMA Surgery, 153(9), 800. doi: 10.1001/jamasurg.2018.1166
- Cowan, D., Bedno, S., Urban, N., Yi, B., & Niebuhr, D. (2011). Musculoskeletal injuries among overweight army trainees: incidence and health care utilization. Occupational Medicine, 61(4), 247-252. doi: 10.1093/occmed/kqr028
- Smith, G. (2000). Hospitalization due to injuries in the military Evaluation of current data and recommendations on their use for injury prevention. American Journal Of Preventive Medicine, 18(1), 41-53. doi: 10.1016/s0749- 3797(99)00171-3
- Lauder, T., Baker, S., Smith, G., & Lincoln, A. (2000). Sports and physical training injury hospitalizations in the Army. American Journal Of Preventive Medicine, 18(1), 118-128. doi: 10.1016/s0749-3797(99)00174-9
- Jones, B., & Hansen, B. (2000). An Armed Forces Epidemiological Board evaluation of injuries in the military. American Journal Of Preventive Medicine, 18(1), 14-25. doi: 10.1016/s0749-3797(99)00170-1
- Cox, P., Kirk, T., Ashmore, T., Willerton, K., Evans, R., & Smith, A., … & Clarke, K. (2016). Nutritional Ketosis Alters Fuel Preference and Thereby Endurance Performance in Athletes. Cell Metabolism, 24(2), 256-268. doi: 10.1016/j.cmet.2016.07.010
- Army Public Health Center. (2017) Health of the Force Report. Retrieved from https://phc.amedd.army.mil/news/Pages/PublicationDetails.aspx?type=Health+of+the+-Force+Report
- National Center for Health Statistics. (2017). Health, United States, 2016


