Assured access to clean and potable water remains a challenge across the globe. In 2018, the United Nations estimated that 1.8 billion people live in regions affected by water scarcity due to land degradation and drought, and more than 4 billion people lack access to safe sanitation [1]. This need is expected to become even more urgent, given current trends in population growth and the consequent need for increased food production [2].
The Department of Defense (DoD) is a major producer and consumer of clean water, both at home and abroad. In fiscal year 2017, DoD facilities and bases worldwide consumed a total of 82.5 billion gallons of potable water (i.e., water purchased from utilities and treated freshwater sources) [3]. However, domestic installations face increasing vulnerability to water scarcity. In July 2018, DoD reported that 21 homeland military installations are located in areas under “severe” or “extreme” drought, as identified by the U.S. Drought Monitor of the National Drought Mitigation Center [3].
As the United Nations’ global drought statistics indicate [1], providing clean and potable water to DoD forces abroad can be a challenge. A major study published in 2010 surveyed water provision to forward operating bases (FOBs) in Iraq and Afghanistan and found that a base camp size of 1,500 warfighters requires at least 13.6 million gallons of water annually—and potentially twice as much [4]. Many FOBs must be supplied with potable water by transportation, which is costly and inefficient [4, 5]. Additionally, the many FOBs that lack wastewater treatment facilities must truck out their wastewater, and FOBs that have treatment facilities often rely on activated sludge processing—which is energy intensive [6].
In order to realize sustainable and effective FOBs, water supply and wastewater treatment methods should be dramatically improved. For these reasons, DoD has sought sustainable and energy-efficient wastewater treatment processes that require less frequent maintenance [7].
Photothermal Water Treatment
Photothermal water treatment has emerged as a promising technique to provide drinking water and increase the working life of pressure-driven membrane filtration. Photothermal materials in a membrane convert incident light, most typically sunlight, into heat by the photothermal effect. Incident photons excite electrons in the photothermal material, which then release their absorbed energy as heat and regain initial state. The locally high temperature produced can inhibit membrane biofouling—a main concern for pressure-driven membrane processes [8, 9]—and it can also promote effective evaporation for clean water generation.
Desirable photothermal membranes exhibit broad-band light absorption across the solar spectrum; have high photothermal conversion rates; and are readily scalable, cost effective, and low in environmental toxicity [10].
This article introduces three of our research group’s recent developments in photothermal water treatment:
- fouling-resistant photothermal membranes in pressure-driven reverse osmosis (RO) and ultrafiltration (UF) systems
- photothermal solar steam generation (SSG) by interfacial evaporators
- photothermal membrane distillation (PMD)
In the anti-fouling membrane, the light-induced high temperature at the membrane surface damaged the cell walls of microorganisms and subsequently inhibited the biofilm growth, which improved the membrane’s durability and performance. In SSG, interfacial solar evaporators achieved high evaporation rates by using engineered nanostructures that are scalable and non-toxic. In PMD, simple and scalable polymeric photothermal materials have been optimized to treat even highly saline water.
Figure 1. Possible photothermal membrane utilization in various DoD installations: (A) Fouling resistant photothermal reverse osmosis and ultrafiltration membrane. Reproduced with permission [16]. Copyright 2019 American Chemical Society. (B) Fixed military installation. (C) Photothermal solar steam generation and photothermal membrane distillation. Reproduced with permission [36]. Copyright 2017 Elsevier. (D) Forward operating bases.
Photothermally-enabled Biofouling Resistance in Membranes for Pressure-driven Filtration
Pressure-driven membrane filtration processes, such as RO and UF, are widely used in DoD installations, owing to their fast water production and large treatment capacities [4, 11]. However, membrane fouling is a persistent challenge in these processes, decreasing the energy efficiency and life span of the membrane, and increasing the cost of water treatment [12]. Among the various types of fouling, biofouling—microorganism sticking to membrane surfaces and forming biofilms—accounts for more than 45% of all membrane fouling and reduces the membrane performance significantly [13]. In the worst case, biofouling contributed to approximately 70% of transmembrane pressure increase in RO [14].
To combat biofouling, we first coupled the photothermal effect with pressure-driven membrane filtration. A commercial polyamide (PA) membrane for RO was surface-modified with gold nanostars (AuNS), graphene oxide (GO), and hydrophilic polyethylene glycol (PEG) (see Figure 2B) [9].
GO nanosheets were employed as templates to grow AuNS in situ, and the AuNS was used to photothermally heat up the membrane surface, as their branched shape can be easily tuned to achieve maximum light absorption in solar spectrum. The localized surface plasmon resonance (LSPR) of AuNS, which involves the collective oscillation of dielectrically confined conduction electrons, can be tuned over the visible to near-infrared (NIR) wavelengths by optimizing the size and aspect ratio of the AuNS branches [15]. The excited electrons relax to ground state, releasing thermal energy (heat). Through photothermal conversion, the PA-GOAuNS-PEG membrane achieved a locally high surface temperature (T= ~70oC) within one minute under 808 nm laser irradiation (see Figure 2C).
This localized heating effectively killed bacteria (e.g., E. coli) on membrane surfaces. In addition to its bactericidal property, the hydrophilic PEG coating on the PA membrane further reduced mineral scaling (CaSO4 and CaCO3) and organic fouling (humic acid), owing to the circumneutral charge and hydrophilic nature of PEG. The newly developed AuNS-GO-PEG-PA membrane reduced organic-, inorganic-, and bio-fouling without any drop in membrane flux or ion rejection efficiency—showing its great promise for adoption in pre-existing pressure-driven membrane filtration processes.
However, AuNS are expensive, and deposited AuNS can be detached from the membrane under high pressure. To overcome this problem, we employed abundant and scalably produced reduced GO (RGO) as a photothermal material and incorporated it into bacterial nanocellulose (BNC), a scalably produced and environmental benign matrix (see Figure 2D) [16]. RGO exhibits broad-band light absorption in the solar spectrum and has high photothermal conversion efficiency due to the facile π-π* transition of loosely held electrons [17]. Thus, the RGO/BNC membrane reached a high surface temperature under simulated sunlight with the same spectrum as sunlight, but three times higher intensity—2.9 kW/m2. Its high temperature inactivated E.coli within three minutes by deteriorating the bacterial cell wall (see Figure 2E).
Moreover, the incorporation of RGO into BNC during bacteria growth results in good mechanical strength without any discernable damage under high pressure (10 bar). Our findings clearly demonstrate that the photothermal membrane under abundant sunlight strongly resisted biofouling, so high membrane performance can be maintained longer, reducing the frequency of membrane replacement. As the DoD possesses numerous membrane filtration units that provide water to warfighters and civilian communities, adapting photothermal membranes in these applications could save energy and reduce the cost of water production.
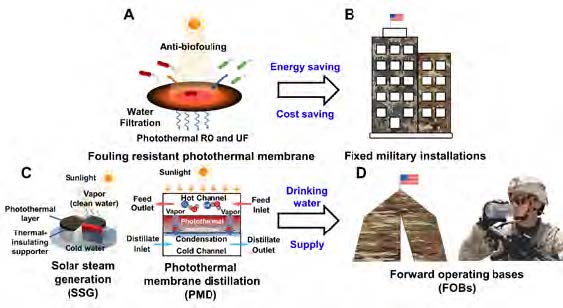
Figure 2. (A) Working principle of photothermal membrane with bio-fouling resistance. Reproduced with permission [16]. Copyright 2019 American Chemical Society. (B) SEM images of PA and PA-GO-AuNS-PEG membranes and TEM image of AuNS. (C) NIR camera images of photothermal membrane under 808 nm laser irradiation (4 kW/m2), showing locally high surface temperature. (B) and (C) are reproduced with permission [9]. Copyright 2015 American Chemical Society. (D) Fabrication of RGO/BNC membrane and its SEM images and photographs. (E) Bactericidal activity against E.coli bacteria on photothermal membrane observed in fluorescence (left) and SEM images (right). (D) and (E) are reproduced with permission [16]. Copyright 2019 American Chemical Society.
Non-pressure-driven Drinking Water Production Using Photothermal Membranes
Although pressure-driven membrane filtration can treat massive volumes of water with high efficiency, it requires large plants to be efficient and it consumes considerable electricity, which may not be available in small FOBs. Most potable water is transported to FOBs at costs ranging from $4.78 to $50.00 per gallon [4]. In this case, SSG and PMD are promising alternatives for supplying clean drinking water because they utilize abundant sunlight as a primary energy source, are efficient even in small scale setups, and have low installation costs [18].
Solar Steam Generation
Solar Steam Generation SSG is direct vaporization of wastewater or saline water by thermal energy from sunlight. The solar evaporator for SSG is composed of a photothermal material with broad-band light absorption as a top surface and a heat insulating supporter as a base [19, 20]. Unlike traditional solar-driven water treatment units, an interfacial solar evaporator with a porous support floats— thus, the heat generated by photothermal effect is confined to the evaporative surface (see Figure 3A) [20]. This locoregional high temperature, together with the controlled amount of water transported by capillary action, promotes evaporation as the evaporation rate is proportional to the temperature at the interface. Considering that SSG requires only sunlight to produce drinkable water and has a low installation cost, it is a promising solution for small FOBs where centralized water treatment is not available. To create cost-effective and efficient solar evaporators, we incorporated well-controlled and functionalized photothermal materials into membranes, making scalable, chemically and mechanically stable, and environmentally-benign photothermal membranes.
First, we developed a RGO/BNC bilayer aerogel as a solar evaporator, composed of a porous BNC supporter and a RGO/ BNC layer on top as the solar absorber (see Figure 3B). Owing to broad-band light absorption of RGO and the heat localization by the thermally insulating BNC layer, the RGO/BNC aerogel achieved high solar evaporation efficiency (~83%) under 10 kW/m2 [20]. Furthermore, the mechanically-interlocked RGO with BNC showed good chemical and mechanical stability. The easy fabrication of these robust RGO/BNC interfacial evaporators makes SSG attractive for real-world applications.
For cost effectiveness and scalability, we introduced two different approaches, using either inexpensive and abundant support (wood) or environmentally-benign photothermal materials (polymerized dopamine [PDA]) [21, 22]. GO was deposited on natural wood by drop casting (see Figure 3C). The hydrophilic vessels in wood transported water from the bottom to the top surface, where the water was effectively vaporized by the photothermal effects of GO under simulated sunlight [21]. These results clearly demonstrated that abundant and inexpensive wood can be used as a supporting layer for solar evaporators.
PDA is a mussel-inspired polymeric material that exhibits strong light absorption and high photothermal conversion efficiency. In PDA synthesis, the monomer, dopamine, can be self-polymerized under alkaline conditions (pH > 7.5) at room temperature without any complicated instruments— making its synthesis scalable and cost effective [23]. Synthesized PDA particles have been incorporated into BNC, similar to RGO/BNC aerogel, and served as a solar evaporator. The fabrication of PDA/BNC aerogel is highly scalable, and the materials are completely biodegradable at the end of its functional life [22].
Molybdenum disulfide (MoS2 ) has emerged as an attractive 2D photothermal material owing to its high light absorptivity in the solar spectrum and its low toxicity [24]. For the first time, we examined the effects of MoS2 phases on light absorption and solar evaporation efficiency [19]. During Li intercalation of the synthesis process, the naturally existing trigonal prismatic crystal phase of MoS2 is transformed to the octahedral phase of MoS2—called chemically exfoliated (ce-) MoS2. The crystal phase change by Li intercalation narrowed the bandgap of MoS2, increasing its solar evaporation efficiency. In addition, the cell toxicity of 2D MoS2 was found to be lower than that of GO with a similar size. Low toxicity and good photothermal conversion efficiency make the ce-MoS2 a promising photothermal material for solar-enabled water treatment and biomedical applications.

Figure 3. (A) Working principle of SSG. Reproduced with permission [36]. Copyright 2017 Elsevier. (B) Fabrication of bilayer solar evaporator using BNC as supporting layer. Reproduced with permission [22]. Copyright 2017 Royal Society of Chemistry. (C) Fabrication of bilayer solar evaporator based on wood. Reproduced with permission [21]. Copyright 2017 American Chemical Society. (D) (Left) Photograph and SEM image of BNC based solar evaporator, AFM and TEM images of photothermal materials (RGO, ce-MoS2, and PDA), and (Right Bottom) photograph of steam generation. Reproduced with permission [19, 20, 22]. Copyright 2018 Elsevier, 2016 Wiley-VCH, and 2017 Royal Society of Chemistry.
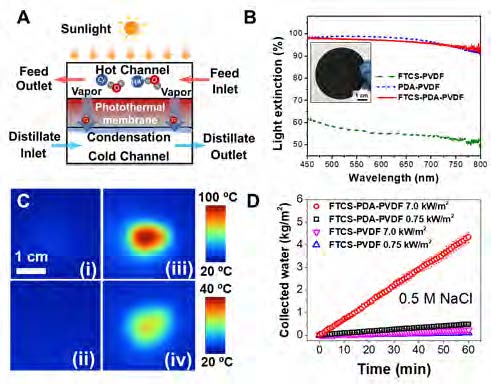
Figure 4. (A) Working principle of photothermal membrane distillation (PMD) process. (B) Light extinction of FTCS–PVDF, PDA–PVDF, and FTCS–PDA–PVDF membranes. Inset: optical image of FTCS–PDA–PVDF membrane. (C) IR camera images of the FTCS–PVDF membrane under irradiations of 7.0 kW m-2 (i) and 0.75 kW m-2 (ii), and the FTCS–PDA–PVDF membrane under 7.0 kW m-2 (iii), and 0.75 kW m-2 (iv), all after 600 seconds illumination. (D) Collected clean water flux of FTCS–PVDF and FTCS–PDA–PVDF membranes when treating 0.5 M NaCl solution at 0.75 kW m-2 and 7.0 kW m-2 using a PMD system. Reproduced with permission [32]. Copyright 2018 Royal Society of Chemistry.
Photothermal Membrane Distillation
Membrane distillation (MD) is a thermally-driven membrane separation process [18]. Unlike pressure-driven membrane filtration, MD can purify water containing high total dissolved solids by using low-grade heat at low pressures, making it less energy intensive [25]. In MD, the temperature difference (∆T) between two sides of a hydrophobic membrane drives vapor-phase water to transport across the membrane [26]. Recently, to utilize solar energy as the main energy source for desalination, MD has been implemented with the aid of photothermal membranes, called PMD [27–29]. In PMD, photothermal materials are coated on the membrane surface (see Figure 4B). The generated heat is localized on the membrane’s top surface, resulting in a higher temperature gradient across the membrane. Furthermore, localized heating by photothermal materials alleviates the temperature polarization that decreases the system efficiency in conventional MD systems [30–32].
To develop an efficient PMD membrane, we synthesized a PDA-coated polyvinylidene fluoride (PVDF) membrane [32]. Facilitated by the outstanding light absorption (Figure 4B) and photothermal conversion properties of PDA (Figure 4C), the membrane achieved high solar conversion efficiency (45%) and a clean water generation flux of 0.49 kg/m2 •h when treating highly saline water (0.5 M NaCl) in a solar-driven direct contact membrane distillation system (Figure 4D) [32]. A simple and easy synthesis method, in situ oxidative polymerization of dopamine on PVDF, makes the membrane highly suitable for potable water generation. Meanwhile, the high salt rejection (> 99.9%) and long-term stability of the membrane make it promising for real-world applications.
Compared to conventional thermal water treatment, our SSG and PMD systems can obtain thermal energy from abundant sunlight to treat wastewater or saline water, making them attractive in FOBs where electricity and potable water are limited. Furthermore, the simple, low-cost setups for SSG and PMD are desirable for small FOBs where drinking water is currently supplied by costly transportation. Thus, these techniques benefit supplying potable water to FOBs by treating gray water produced on site, reducing the amounts of wastewater.
Conclusion
The newly-developed photothermal membranes are an appealing way to provide safe, clean drinking water. They can be used in both large-scale water treatment plants for fixed military installations and small-scale water treatment facilities at FOBs. However, translating these laboratory scale studies into portable devices and utility-scale plants without compromising efficiency remains a challenge.
To exploit the anti-biofouling property of photothermal membranes, the conventional spiral-wound shape modules in closed RO/UF systems will need to be redesigned for sunlight accessibility [16]. In SSG, water droplets formed on the condensation surface can reflect and scatter light, reducing the light absorption by the evaporator [33]. In the future, improved steam condensation and collection systems should be developed to maximize the clean water generation rate. Future PMD systems should achieve improved water generation rates and solar conversion efficiencies. New modules with multilayer heat recovery setups can improve the overall energy efficiency of PMD [34, 35]. Combining PMD with low grade heat energy sources, (e.g., engine cooling waters and waste incineration), can further increase its water production rate. For both SSG and PMD, portable and modular devices need to be developed to generate potable water for FOBs.
Acknowledgments
The authors acknowledge support from National Science Foundation Environmental Engineering Program (CBET-1604542), and Professor James C. Ballard for carefully reviewing the manuscript. Also, the authors thank our current and previous group members, especially Dr. Qisheng Jiang, Mr. Hamed Gholami Derami, Dr. Keng-Ku Liu, and Professor Jessica R. Ray, for their valuable findings which contributed a lot for this paper.
References
- United Nations Educational, Scientific, and Cultural Organization. (2018). The United Nations World Water Development Report 2018: Nature-Based Solutions for Water. Retrieved from https://unesdoc.unesco.org/ ark:/48223/pf0000261424
- Mekonnen, M. M., & Hoekstra, A. Y. (2016). Four billion people facing severe water scarcity. Science Advances (2)2, e1500323. doi:10.1126/sciadv.1500323
- Department of Defense. (2018, July). Annual Energy Management and Resilience Report (AEMRR), Fiscal Year 2017. Office of the Assistant Secretary of Defense for Energy, Installations, and Environment. Retrieved from https://www.acq.osd.mil/eie/ Downloads/IE/FY%202017%20AEMR.pdf
- Noblis. (2010, May 21). Sustainable forward operating bases. White Paper, Strategic Environmental Research and Development Program (SERDP). Retrieved from https:// apps.dtic.mil/dtic/tr/fulltext/u2/a571503.pdf
- Anderson, H. G., Maloney, S. W., Kinnevan, K., Smith, E. D., Hay, J., & Gerdes, G. L. (2013, September). Baseline water demand at forward operating bases. U.S. Army Corps of Engineers, Engineer Research and Development Center (ERDC). Retrieved from https://apps.dtic.mil/dtic/tr/fulltext/u2/ a613424.pdf
- Guy, K. A., Page, M. A., Schieman, L., & Botte, G. (2016, August). Novel anaerobic wastewater treatment system for energy generation at forward operating bases: Final report on Envionmental Restoration Project ER-2218. U.S. Army Corps of Engineers, Engineer Research and Development Center (ERDC). Retrieved from https://apps.dtic. mil/dtic/tr/fulltext/u2/1014910.pdf
- Environmental Security Technology Certification Program. (2019, Februay 8). ESTCP announces FY 2019 new start project selections. Depatment of Defense. Retrieved from https://www.serdp-estcp. org/News-and-Events/News-Announcements/Program-News/ESTCP-announces-FY-2019-new-start-project-selections
- Ghasemi, H., Ni, G., Marconnet, A. M., Loomis, J., Yerci, S., Miljkovic, N., & Chen, G. (2014). Solar steam generation by heat localization. Nature Communications, 5(1), 4449. doi:10.1038/ncomms5449
- Ray, J. R., Tadepalli, S., Nergiz, S. Z., Liu, K., You, L., Tang, Y., . . . Jun, Y. (2015). Hydrophilic, bactericidal nanoheater-enabled reverse osmosis membranes to improve fouling resistance. ACS Applied Materials & Interfaces, 7(21), 11117–11126. doi:10.1021/ am509174j
- Shi, Y., Li, R., Jin, Y., Zhuo, S., Shi, L., Chang, J., . . . Wang, P. (2018). A 3D photothermal structure toward improved energy efficiency in solar steam generation. Joule, 2(6), 1171–1186. doi:10.1016/j. joule.2018.03.013
- Elimelech, M., & Phillip, W. A. (2011). The future of seawater desalination: Energy, technology, and the environment. Science, 333(6043), 712–717. doi:10.1126/science.1200488
- Zhang, R., Liu, Y., He, M., Su, Y., Zhao, X., Elimelech, M., & Jiang, Z. (2016). Antifouling membranes for sustainable water purification: Strategies and mechanisms. Chemical Society Reviews, 45(21), 5888–5924. doi:10.1039/c5cs00579e
- Flemming, H., Schaule, G., Griebe, T., Schmitt, J., & Tamachkiarowa, A. (1997). Biofouling—The Achilles heel of membrane processes. Desalination, 113(2-3), 215–225. doi:10.1016/s0011-9164(97)00132-x
- Herzberg, M., & Elimelech, M. (2007). Biofouling of reverse osmosis membranes: Role of biofilm-enhanced osmotic pressure. Journal of Membrane Science, 295(1-2), 11–20. doi:10.1016/j.memsci.2007.02.024
- Hao, F., Nehl, C. L., Hafner, J. H., & Nordlander, P. (2007). Plasmon Resonances of a gold nanostar. Nano Letters, 7(3), 729–732. doi:10.1021/nl062969c
- Jiang, Q., Ghim, D., Cao, S., Tadepalli, S., Liu, K., Kwon, H., . . . Singamaneni, S. (2019). Photothermally active reduced graphene oxide/bacterial nanocellulose composites as biofouling-resistant ultrafiltration membranes. Environmental Science & Technology, 53(1), 412–421. doi:10.1021/ acs.est.8b02772
- Bond, T. C., & Bergstrom, R. W. (2006). Light absorption by carbonaceous particles: An investigative review. Aerosol Science and Technology, 40(1), 27–67. doi:10.1080/02786820500421521
- Deshmukh, A., Boo, C., Karanikola, V., Lin, S., Straub, A. P., Tong, T., . . . Elimelech, M. (2018). Membrane distillation at the water-energy nexus: Limits, opportunities, and challenges. Energy & Environmental Science, 11(5), 1177–1196. doi:10.1039/ c8ee00291f
- Ghim, D., Jiang, Q., Cao, S., Singamaneni, S., & Jun, Y. (2018). Mechanically interlocked 1T/2H phases of MoS2 nanosheets for solar thermal water purification. Nano 26 Energy, 53, 949–957. doi:10.1016/j.nanoen.2018.09.038
- Jiang, Q., Tian, L., Liu, K., Tadepalli, S., Raliya, R., Biswas, P., . . . Singamaneni, S. (2016). Bilayered biofoam for Highly efficient solar steam generation. Advanced Materials, 28(42), 9400–9407. doi:10.1002/ adma.201601819
- Liu, K., Jiang, Q., Tadepalli, S., Raliya, R., Biswas, P., Naik, R. R., & Singamaneni, S. (2017). Wood–graphene oxide composite for highly efficient solar steam generation and desalination. ACS Applied Materials & Interfaces, 9(8), 7675–7681. doi:10.1021/ acsami.7b01307
- Jiang, Q., Derami, H. G., Ghim, D., Cao, S., Jun, Y., & Singamaneni, S. (2017). Polydopamine-filled bacterial nanocellulose as a biodegradable interfacial photothermal evaporator for highly efficient solar steam generation. Journal of Materials Chemistry A, 5(35), 18397–18402. doi:10.1039/ c7ta04834c
- Liu, Y., Ai, K., & Lu, L. (2014). Polydopamine and its derivative materials: Synthesis and promising applications in energy, environmental, and biomedical fields. Chemical Reviews, 114(9), 5057–5115. doi:10.1021/ cr400407a
- Wang, Z., & Mi, B. (2017). Environmental applications of 2D molybdenum disulfide (MoS2) nanosheets. Environmental Science & Technology, 51(15), 8229–8244. doi:10.1021/acs.est.7b01466
- Camacho, L. M., Dumée, L., Zhang, J., Li, J., Duke, M., Gomez, J., & Gray, S. (2013). Advances in membrane distillation for water desalination and purification applications. Water, 5(1), 94–196. doi:10.3390/w5010094
- Alkhudhiri, A., Darwish, N., & Hilal, N. (2012). Membrane distillation: A comprehensive review. Desalination, 287, 2–18. doi:10.1016/j.desal.2011.08.027
- Ahmed, F. E., Hashaikeh, R., & Hilal, N. (2019). Solar powered desalination – Technology, energy and future outlook. Desalination, 453, 54–76. doi:10.1016/j.desal.2018.12.002
- Wang, P. (2018). Emerging investigator series: The rise of nano-enabled photothermal materials for water evaporation and clean water production by sunlight. Environmental Science: Nano, 5(5), 1078–1089. doi:10.1039/c8en00156a
- Westerhoff, P., Alvarez, P., Li, Q., Gardea-Torresdey, J., & Zimmerman, J. (2016). Overcoming implementation barriers for nanotechnology in drinking water treatment. Environmental Science: Nano, 3(6), 1241– 1253. doi:10.1039/c6en00183a
- Dongare, P. D., Alabastri, A., Pedersen, S., Zodrow, K. R., Hogan, N. J., Neumann, O., . . . Halas, N. J. (2017). Nanophotonics-enabled solar membrane distillation for off-grid water purification. Proceedings of the National Academy of Sciences, 114(27), 6936–6941. doi:10.1073/pnas.1701835114
- Politano, A., Argurio, P., Profio, G. D., Sanna, V., Cupolillo, A., Chakraborty, S., . . . Curcio, E. (2016). Photothermal membrane distillation for seawater Desalination. Advanced Materials, 29(2), 1603504. doi:10.1002/adma.201603504
- Wu, X., Jiang, Q., Ghim, D., Singamaneni, S., & Jun, Y. (2018). Localized heating with a photothermal polydopamine coating facilitates a novel membrane distillation process. Journal of Materials Chemistry A, 6(39), 18799–18807. doi:10.1039/c8ta05738a
- Ni, G., Zandavi, S. H., Javid, S. M., Boriskina, S. V., Cooper, T. A., & Chen, G. (2018). A salt-rejecting floating solar still for lowcost desalination. Energy & Environmental Science, 11(6), 1510–1519. doi:10.1039/ c8ee00220g
- Chiavazzo, E., Morciano, M., Viglino, F., Fasano, M., & Asinari, P. (2018). Passive solar high-yield seawater desalination by modular and low-cost distillation. Nature Sustainability, 1(12), 763–772. doi:10.1038/s41893- 018-0186-x
- Xue, G., Chen, Q., Lin, S., Duan, J., Yang, P., Liu, K., . . . Zhou, J. (2018). Highly efficient water harvesting with optimized solar thermal membrane distillation device. Global Challenges, 2(5-6), 1800001. doi:10.1002/ gch2.201800001
- Jiang, Q., & Singamaneni, S. (2017). Water from wood: Pouring through pores. Joule, 1(3), 429–430. doi:10.1016/j. joule.2017.10.018


